| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:14:20 UTC |
|---|
| Update Date | 2016-11-09 01:17:45 UTC |
|---|
| Accession Number | CHEM023698 |
|---|
| Identification |
|---|
| Common Name | alpha-N-Carboethoxyacetyl-4-chloro-D-tryptophan |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
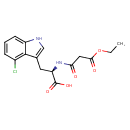
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2R)-3-(4-Chloro-1H-indol-3-yl)-2-[(3-ethoxy-1-hydroxy-3-oxopropylidene)amino]propanoate | Generator | | a-N-Carboethoxyacetyl-4-chloro-D-tryptophan | Generator | | Α-N-carboethoxyacetyl-4-chloro-D-tryptophan | Generator |
|
|---|
| Chemical Formula | C16H17ClN2O5 |
|---|
| Average Molecular Mass | 352.770 g/mol |
|---|
| Monoisotopic Mass | 352.083 g/mol |
|---|
| CAS Registry Number | 27564-24-5 |
|---|
| IUPAC Name | (2R)-3-(4-chloro-1H-indol-3-yl)-2-(3-ethoxy-3-oxopropanamido)propanoic acid |
|---|
| Traditional Name | (2R)-3-(4-chloro-1H-indol-3-yl)-2-(3-ethoxy-3-oxopropanamido)propanoic acid |
|---|
| SMILES | CCOC(=O)CC(=O)N[C@H](CC1=CNC2=CC=CC(Cl)=C12)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C16H17ClN2O5/c1-2-24-14(21)7-13(20)19-12(16(22)23)6-9-8-18-11-5-3-4-10(17)15(9)11/h3-5,8,12,18H,2,6-7H2,1H3,(H,19,20)(H,22,23)/t12-/m1/s1 |
|---|
| InChI Key | DWVLUSJVGYOTDB-GFCCVEGCSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as n-acyl-alpha amino acids. N-acyl-alpha amino acids are compounds containing an alpha amino acid which bears an acyl group at its terminal nitrogen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | N-acyl-alpha amino acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - N-acyl-alpha-amino acid
- 3-alkylindole
- Indole
- Indole or derivatives
- Aryl chloride
- Aryl halide
- Dicarboxylic acid or derivatives
- Benzenoid
- 1,3-dicarbonyl compound
- Substituted pyrrole
- Heteroaromatic compound
- Pyrrole
- Carboxamide group
- Carboxylic acid ester
- Secondary carboxylic acid amide
- Carboxylic acid
- Azacycle
- Organoheterocyclic compound
- Organooxygen compound
- Organic oxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Carbonyl group
- Organohalogen compound
- Organochloride
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-2469000000-d13770acb99ea0a79edf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000f-4972000000-55f16906d59c88b8df9e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03di-2910000000-c70f96472d3bf355d986 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0zfr-2229000000-ed0e289e9d22b79bbdc2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052s-7595000000-66e6c46d80fd0c0c0d36 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052v-9240000000-72448a6114ba2f86ef6c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0079-0093000000-f3d96fb1fe2353c0104f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dr-1391000000-d7224c8fb575bb874196 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6y-4920000000-ec6438dfb1bf30d2a68e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-2249000000-829f7931523f827e42e7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f89-9721000000-69cd61140764b4fd50c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03di-7940000000-716933ac643a10171832 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0301784 |
|---|
| FooDB ID | FDB001406 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 59696182 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 154350626 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|