| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:12:30 UTC |
|---|
| Update Date | 2016-11-09 01:17:45 UTC |
|---|
| Accession Number | CHEM023649 |
|---|
| Identification |
|---|
| Common Name | Euglobal IVa |
|---|
| Class | Small Molecule |
|---|
| Description | Euglobal IVb is a constituent of Eucalyptus globulus (Tasmanian blue gum) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
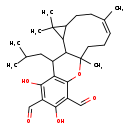
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (±)-6-methyl-5-hepten-2-yl acetic acid | HMDB | | 1,5-Dimethylhex-4-enyl acetate | HMDB | | 5-Hepten-2-ol, 6-methyl-, 2-acetate | HMDB | | 5-Hepten-2-ol, 6-methyl-, acetate | HMDB | | 6-Methyl-5-hepten-2-ol, acetate | HMDB |
|
|---|
| Chemical Formula | C28H38O5 |
|---|
| Average Molecular Mass | 454.598 g/mol |
|---|
| Monoisotopic Mass | 454.272 g/mol |
|---|
| CAS Registry Number | 77794-65-1 |
|---|
| IUPAC Name | (7Z)-15,17-dihydroxy-3,3,7,11-tetramethyl-19-(2-methylpropyl)-12-oxatetracyclo[9.8.0.0²,⁴.0¹³,¹⁸]nonadeca-7,13(18),14,16-tetraene-14,16-dicarbaldehyde |
|---|
| Traditional Name | (7Z)-15,17-dihydroxy-3,3,7,11-tetramethyl-19-(2-methylpropyl)-12-oxatetracyclo[9.8.0.0²,⁴.0¹³,¹⁸]nonadeca-7,13(18),14,16-tetraene-14,16-dicarbaldehyde |
|---|
| SMILES | CC(C)CC1C2C3C(CC\C(C)=C/CCC2(C)OC2=C1C(O)=C(C=O)C(O)=C2C=O)C3(C)C |
|---|
| InChI Identifier | InChI=1S/C28H38O5/c1-15(2)12-17-21-25(32)18(13-29)24(31)19(14-30)26(21)33-28(6)11-7-8-16(3)9-10-20-23(22(17)28)27(20,4)5/h8,13-15,17,20,22-23,31-32H,7,9-12H2,1-6H3/b16-8- |
|---|
| InChI Key | XJFLMCYKZVYATJ-PXNMLYILSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 1-benzopyrans. These are organic aromatic compounds that 1-benzopyran, a bicyclic compound made up of a benzene ring fused to a pyran, so that the oxygen atom is at the 1-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzopyrans |
|---|
| Sub Class | 1-benzopyrans |
|---|
| Direct Parent | 1-benzopyrans |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1-benzopyran
- Aryl-aldehyde
- Alkyl aryl ether
- Benzenoid
- Vinylogous acid
- Oxacycle
- Ether
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Aldehyde
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-01p6-4003900000-7ad70b5b22b38f6d3fb9 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-001i-2400090000-c35053c3805935932407 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0020900000-6df94a6be302bb301db2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4r-4053900000-3c1d47ed2e5a0587f258 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-1009-9342200000-3df50008cdc79c09482a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000900000-1f326c781a0d0ebc2cc4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0000900000-11bf6179c10a0bf41ba8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-02cl-3249700000-ad1325ef396b7bf73e7d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0001900000-cb111fe5f8e419e81be0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0bt9-0011900000-ae95e0e948dd2fd0b70f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gvo-5439500000-5bea6f4371d01557a2c1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000900000-0db53eae8610a0e47ec3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0000900000-af4793f0397f2a7fe3fb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f83-4097800000-dfc45b1499122dfcd997 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030035 |
|---|
| FooDB ID | FDB001336 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013119 |
|---|
| ChEBI ID | 175628 |
|---|
| PubChem Compound ID | 131750946 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|