| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 21:07:43 UTC |
|---|
| Update Date | 2016-11-09 01:17:44 UTC |
|---|
| Accession Number | CHEM023536 |
|---|
| Identification |
|---|
| Common Name | Gein |
|---|
| Class | Small Molecule |
|---|
| Description | Sasanquin is found in fats and oils. Sasanquin is isolated from leaves of Camellia sasanqua and Camellia japonica. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
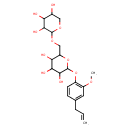
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3,7-Dimethyl-1-(2-propynyl)xanthine | HMDB | | 3,7-Dimethyl-1-propargylxanthine | HMDB | | 3,7-Dimethyl-I-propargylxanthine | HMDB | | DMPX | HMDB | | Eugenol vicanoside | HMDB | | Geoside | HMDB |
|
|---|
| Chemical Formula | C21H30O11 |
|---|
| Average Molecular Mass | 458.456 g/mol |
|---|
| Monoisotopic Mass | 458.179 g/mol |
|---|
| CAS Registry Number | 585-90-0 |
|---|
| IUPAC Name | 2-[2-methoxy-4-(prop-2-en-1-yl)phenoxy]-6-{[(3,4,5-trihydroxyoxan-2-yl)oxy]methyl}oxane-3,4,5-triol |
|---|
| Traditional Name | 2-[2-methoxy-4-(prop-2-en-1-yl)phenoxy]-6-{[(3,4,5-trihydroxyoxan-2-yl)oxy]methyl}oxane-3,4,5-triol |
|---|
| SMILES | COC1=C(OC2OC(COC3OCC(O)C(O)C3O)C(O)C(O)C2O)C=CC(CC=C)=C1 |
|---|
| InChI Identifier | InChI=1S/C21H30O11/c1-3-4-10-5-6-12(13(7-10)28-2)31-21-19(27)17(25)16(24)14(32-21)9-30-20-18(26)15(23)11(22)8-29-20/h3,5-7,11,14-27H,1,4,8-9H2,2H3 |
|---|
| InChI Key | FSCNUJMKSQHQSY-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenolic glycosides. These are organic compounds containing a phenolic structure attached to a glycosyl moiety. Some examples of phenolic structures include lignans, and flavonoids. Among the sugar units found in natural glycosides are D-glucose, L-Fructose, and L rhamnose. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Phenolic glycosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenolic glycoside
- Disaccharide
- O-glycosyl compound
- Anisole
- Phenoxy compound
- Phenol ether
- Methoxybenzene
- Alkyl aryl ether
- Oxane
- Monocyclic benzene moiety
- Benzenoid
- Secondary alcohol
- Polyol
- Ether
- Organoheterocyclic compound
- Acetal
- Oxacycle
- Hydrocarbon derivative
- Alcohol
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-01oy-4586900000-250014060847faef5d6f | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-0bta-3233219000-277675539b8c0b7db716 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-066u-0901500000-100838ec2d480fa70753 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0901000000-446e16cec3a480e68261 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00kb-2900000000-b7f06d168ba046afd03c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-08fr-1922600000-d0c473b49bbd64ffdf04 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03ea-0900100000-84bd55d596a54f7a272b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01ot-2900000000-e0c04600ea5548a42cef | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4l-0513900000-5a763d7ae727c57c8062 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05o0-0901100000-370d47beb94572e7148c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-1901000000-3aae5354806919f8eade | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0btj-0902500000-913b0ead288934c9a837 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-016r-1509000000-b37ff1c22ebbfff89adf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bta-2900200000-72d28f63a33d8e862fa9 | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0029909 |
|---|
| FooDB ID | FDB001162 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00030363 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Ed Gein |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 168209 |
|---|
| PubChem Compound ID | 12310056 |
|---|
| Kegg Compound ID | C17058 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|