| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 20:47:00 UTC |
|---|
| Update Date | 2016-11-09 01:17:37 UTC |
|---|
| Accession Number | CHEM023014 |
|---|
| Identification |
|---|
| Common Name | Conidendrin |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
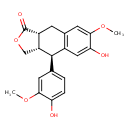
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| alpha-Conidendrin | MeSH |
|
|---|
| Chemical Formula | C20H20O6 |
|---|
| Average Molecular Mass | 356.369 g/mol |
|---|
| Monoisotopic Mass | 356.126 g/mol |
|---|
| CAS Registry Number | 518-55-8 |
|---|
| IUPAC Name | (3aS,4R,9aR)-6-hydroxy-4-(4-hydroxy-3-methoxyphenyl)-7-methoxy-1H,3H,3aH,4H,9H,9aH-naphtho[2,3-c]furan-1-one |
|---|
| Traditional Name | (3aS,4R,9aR)-6-hydroxy-4-(4-hydroxy-3-methoxyphenyl)-7-methoxy-3H,3aH,4H,9H,9aH-naphtho[2,3-c]furan-1-one |
|---|
| SMILES | COC1=CC2=C(C=C1O)[C@H]([C@@H]1COC(=O)[C@@H]1C2)C1=CC=C(O)C(OC)=C1 |
|---|
| InChI Identifier | InChI=1S/C20H20O6/c1-24-17-6-10(3-4-15(17)21)19-12-8-16(22)18(25-2)7-11(12)5-13-14(19)9-26-20(13)23/h3-4,6-8,13-14,19,21-22H,5,9H2,1-2H3/t13-,14-,19-/m1/s1 |
|---|
| InChI Key | CAYMSCGTKZIVTN-PJIJBLCYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as lignan lactones. These are lignans that contain a lactone moiety. They include 1-aryltetralin lactones, dibenzylbutyrolactone lignans, and podophyllotoxins, among others. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lignans, neolignans and related compounds |
|---|
| Class | Lignan lactones |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Lignan lactones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Lignan lactone
- 1-aryltetralin lignan
- Naphthofuran
- Methoxyphenol
- Tetralin
- Phenoxy compound
- Anisole
- Methoxybenzene
- Phenol ether
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- Phenol
- Monocyclic benzene moiety
- Gamma butyrolactone
- Benzenoid
- Tetrahydrofuran
- Lactone
- Carboxylic acid ester
- Carboxylic acid derivative
- Oxacycle
- Ether
- Organoheterocyclic compound
- Monocarboxylic acid or derivatives
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0019000000-cd3bb8d5a58762e04801 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-3179000000-5d67cc69fe3af5717e34 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00os-0492000000-f6c1882495d0e7c92d0e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0009000000-adf41a8fd4548aedb025 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0bt9-0019000000-0fe76f1003a1996b17eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4j-0096000000-6bd31d1773ac0b777c3b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0009000000-bec38414c6d4e5f8a1b4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0159000000-412cdc0cf39e4bee70be | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03dr-0297000000-4cd8ee90ba05049ca5a3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0009000000-0a6526e745d11b23421e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4r-0109000000-9c44c451d41e69179b99 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a7s-0089000000-14eaa4a8fc33e6c2b5e4 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0301742 |
|---|
| FooDB ID | FDB000331 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 65417 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 72506 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|