| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 20:45:27 UTC |
|---|
| Update Date | 2016-11-09 01:17:36 UTC |
|---|
| Accession Number | CHEM022958 |
|---|
| Identification |
|---|
| Common Name | Ferulic acid 4-glucoside |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
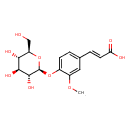
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2E)-3-(3-Methoxy-4-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)prop-2-enoate | Generator | | Ferulate 4-glucoside | Generator | | (2E)-3-[4-(beta-D-Glucopyranosyloxy)-3-methoxyphenyl]-2-propenoic acid | PhytoBank | | (2E)-3-[4-(β-D-Glucopyranosyloxy)-3-methoxyphenyl]-2-propenoic acid | PhytoBank | | (E)-Ferulic acid 4-O-beta-D-glucoside | PhytoBank | | (E)-Ferulic acid 4-O-β-D-glucoside | PhytoBank | | Ferulic acid 4-O-glucoside | PhytoBank | | Ferulic acid 4-O-beta-glucopyranoside | PhytoBank | | Ferulic acid 4-O-β-glucopyranoside | PhytoBank | | Lavandoside | PhytoBank | | trans-4-O-beta-D-Glucopyranosylferulic acid | PhytoBank | | trans-4-O-β-D-Glucopyranosylferulic acid | PhytoBank | | trans-Ferulic acid 4-beta-D-glucoside | PhytoBank | | trans-Ferulic acid 4-β-D-glucoside | PhytoBank | | trans-Ferulic acid-4-beta-glucoside | PhytoBank | | trans-Ferulic acid-4-β-glucoside | PhytoBank | | trans-p-Feruloyl-beta-D-glucopyranoside | PhytoBank | | trans-p-Feruloyl-β-D-glucopyranoside | PhytoBank | | Ferulic acid 4-O-beta-D-glucopyranoside | PhytoBank | | Ferulic acid 4-O-β-D-glucopyranoside | PhytoBank |
|
|---|
| Chemical Formula | C16H20O9 |
|---|
| Average Molecular Mass | 356.325 g/mol |
|---|
| Monoisotopic Mass | 356.111 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2E)-3-(3-methoxy-4-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)prop-2-enoic acid |
|---|
| Traditional Name | (2E)-3-(3-methoxy-4-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)prop-2-enoic acid |
|---|
| SMILES | COC1=CC(\C=C\C(O)=O)=CC=C1O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C16H20O9/c1-23-10-6-8(3-5-12(18)19)2-4-9(10)24-16-15(22)14(21)13(20)11(7-17)25-16/h2-6,11,13-17,20-22H,7H2,1H3,(H,18,19)/b5-3+/t11-,13-,14+,15-,16-/m1/s1 |
|---|
| InChI Key | IEMIRSXOYFWPFD-BJGSYIFTSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenolic glycosides. These are organic compounds containing a phenolic structure attached to a glycosyl moiety. Some examples of phenolic structures include lignans, and flavonoids. Among the sugar units found in natural glycosides are D-glucose, L-Fructose, and L rhamnose. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Phenolic glycosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenolic glycoside
- Cinnamic acid
- Cinnamic acid or derivatives
- Coumaric acid or derivatives
- Hexose monosaccharide
- O-glycosyl compound
- Phenoxy compound
- Anisole
- Methoxybenzene
- Styrene
- Phenol ether
- Alkyl aryl ether
- Monocyclic benzene moiety
- Benzenoid
- Monosaccharide
- Oxane
- Secondary alcohol
- Monocarboxylic acid or derivatives
- Ether
- Organoheterocyclic compound
- Oxacycle
- Carboxylic acid
- Carboxylic acid derivative
- Acetal
- Polyol
- Alcohol
- Carbonyl group
- Organic oxide
- Primary alcohol
- Hydrocarbon derivative
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-054t-0916000000-ebc1cc4896fdc64a458c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-002b-0900000000-6a902dabbbf78442f23e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004s-1900000000-e4ad1acd6f4850f0bc2e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4l-1819000000-eba6f8f85bf8d5d53934 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-002f-1911000000-77f2053078c1eb574d28 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004l-2900000000-2ff1d99e129350f50621 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052s-0219000000-b7234cdb53b60902464c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01y2-0914000000-a18d425b2a03a2eee99b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001j-1920000000-3cd8ce35afe706d70b23 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0109000000-be106f09d2f43d279f4f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-2893000000-9a4389f84bbaed564315 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0002-3902000000-98c283edf136c4ee19bf | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0301717 |
|---|
| FooDB ID | FDB000256 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 10212167 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|