| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:55:13 UTC |
|---|
| Update Date | 2016-11-09 01:17:35 UTC |
|---|
| Accession Number | CHEM022822 |
|---|
| Identification |
|---|
| Common Name | N-Acetylmannosamine |
|---|
| Class | Small Molecule |
|---|
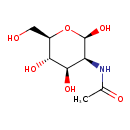
| Description | N-Acetylmannosamine, also known as n-acetylmannosamine or n-acetylmannosamine, belongs to the class of organic compounds known as acylaminosugars. These are organic compounds containing a sugar linked to a chain through N-acyl group. N-Acetylmannosamine is possibly soluble (in water) and an extremely weak basic (essentially neutral) compound (based on its pKa). N-Acetylmannosamine participates in a number of enzymatic reactions, within cattle. In particular, N-Acetylmannosamine can be biosynthesized from N-acetyl-D-glucosamine; which is catalyzed by the enzyme N-acylglucosamine 2-epimerase. In addition, N-Acetylmannosamine and uridine 5'-diphosphate can be biosynthesized from uridine diphosphate-N-acetylglucosamine through the action of the enzyme bifunctional UDP-N-acetyl glucosamine 2-epimerase / N-acetylmannosamine kinase. In cattle, N-acetylmannosamine is involved in the metabolic pathway called the amino sugar metabolism pathway. |
|---|
| Contaminant Sources | - FooDB Chemicals
- HMDB Contaminants - Feces
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| beta-ManNAc | ChEBI | | N-Acetyl-beta-mannosamine | ChEBI | | b-ManNAc | Generator | | Β-mannac | Generator | | N-Acetyl-b-mannosamine | Generator | | N-Acetyl-β-mannosamine | Generator | | 2-Acetamido-2-deoxy-D-mannopyranose | HMDB | | 2-Acetamido-2-deoxy-D-mannose | HMDB | | ManNAc | HMDB | | N-Acetyl-D-mannosamine | HMDB | | N-Acetylmannosamine, (D)-isomer | HMDB | | N-Acetylmannosamine, (L)-isomer | HMDB |
|
|---|
| Chemical Formula | C8H15NO6 |
|---|
| Average Molecular Mass | 221.208 g/mol |
|---|
| Monoisotopic Mass | 221.090 g/mol |
|---|
| CAS Registry Number | 3615-17-6 |
|---|
| IUPAC Name | N-[(2R,3S,4R,5S,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)oxan-3-yl]acetamide |
|---|
| Traditional Name | N-acetylmannosamine |
|---|
| SMILES | [H][C@@]1(O)O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1NC(C)=O |
|---|
| InChI Identifier | InChI=1S/C8H15NO6/c1-3(11)9-5-7(13)6(12)4(2-10)15-8(5)14/h4-8,10,12-14H,2H2,1H3,(H,9,11)/t4-,5+,6-,7-,8-/m1/s1 |
|---|
| InChI Key | OVRNDRQMDRJTHS-OZRXBMAMSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as acylaminosugars. These are organic compounds containing a sugar linked to a chain through N-acyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Acylaminosugars |
|---|
| Alternative Parents | |
|---|
| Substituents | - Acylaminosugar
- N-acyl-alpha-hexosamine
- Hexose monosaccharide
- Monosaccharide
- Oxane
- Acetamide
- Carboxamide group
- Hemiacetal
- Secondary carboxylic acid amide
- Secondary alcohol
- Carboxylic acid derivative
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Organic nitrogen compound
- Organopnictogen compound
- Organonitrogen compound
- Primary alcohol
- Carbonyl group
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| GC-MS | GC-MS Spectrum - GC-MS (4 TMS) | splash10-00di-1910000000-c54e9f9969c7d963036f | Spectrum | | GC-MS | GC-MS Spectrum - GC-MS (1 MEOX; 5 TMS) | splash10-0pvi-1951000000-56398608d2dd4355e1f6 | Spectrum | | GC-MS | GC-MS Spectrum - GC-MS (1 MEOX; 4 TMS) | splash10-0kw0-3940000000-02993315f1b1a700b7ec | Spectrum | | GC-MS | GC-MS Spectrum - GC-MS (1 MEOX; 5 TMS) | splash10-0pvi-2941000000-c2e3ada4d3e27dc6890a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udl-7920000000-9991e74281c314048f5e | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (4 TMS) - 70eV, Positive | splash10-0007-3331900000-c6fab10d67507eecce65 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-004r-0920000000-26c87e12e0c8c5f919b7 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-001i-9400000000-f0d2f81f4292b627f0ef | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-001j-9000000000-fcbf9adb619f826e3cd3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0390000000-ce54c5d356ec2fc8ab9f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0il0-2950000000-e4e02ffea74d8dfd0810 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01ox-9400000000-180503dcc7682bb1753b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00dr-9830000000-a5028d14de09cabbb303 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ac0-9820000000-d3017a9fb13bdc3aa847 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-9200000000-ce24407926ce180ba365 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0390000000-3820a7eec5a8c72f49d9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0h90-9740000000-db880363194a3979c637 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0229-9200000000-cdcf015aa799dc7d6d8b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9500000000-2c2b09cb6bfb29039085 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0abc-9540000000-a1f7d557237a521d306a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-18eabe9e3d3657888360 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 2D NMR | [1H,1H] 2D NMR Spectrum | Not Available | Spectrum | | 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0001129 |
|---|
| FooDB ID | FDB028419 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | 35606 |
|---|
| BioCyc ID | N-ACETYL-D-MANNOSAMINE |
|---|
| METLIN ID | 6024 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | N-acetylmannosamine |
|---|
| Chemspider ID | 9271300 |
|---|
| ChEBI ID | 63154 |
|---|
| PubChem Compound ID | 11096158 |
|---|
| Kegg Compound ID | C00645 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Satoh, Chiyoko; Akio, Kiyomoto. Nitrogen-containing sugars. I. Synthesis of 2-acetamido-2-deoxy-D-mannose from 1-deoxy-1-nitro-D-mannitol pentaacetate. Chemical & Pharmaceutical Bulletin (1964), 12(5), 615-19. | | 2. Sugawara T, Irie K, Iwasawa H, Yoshikawa T, Okuno S, Watanabe HK, Kato T, Shibukawa M, Ito Y: Synthesis of omega-(methoxycarbonyl)alkyl and 9-(methoxycarbonyl)-3,6-dioxanonyl glycopyranosides for the preparation of carbohydrate-protein conjugates. Carbohydr Res. 1992 Jun 4;230(1):117-49. | | 3. Salama I, Hinderlich S, Shlomai Z, Eisenberg I, Krause S, Yarema K, Argov Z, Lochmuller H, Reutter W, Dabby R, Sadeh M, Ben-Bassat H, Mitrani-Rosenbaum S: No overall hyposialylation in hereditary inclusion body myopathy myoblasts carrying the homozygous M712T GNE mutation. Biochem Biophys Res Commun. 2005 Mar 4;328(1):221-6. | | 4. Kamerling JP, Strecker G, Farriaux JP, Dorland L, Haverkamp J, Vliegenthart JF: 2-Acetamidoglucal, a new metabolite isolated from the urine of a patient with sialuria. Biochim Biophys Acta. 1979 Mar 22;583(3):403-8. | | 5. Sala G, Dupre T, Seta N, Codogno P, Ghidoni R: Increased biosynthesis of glycosphingolipids in congenital disorder of glycosylation Ia (CDG-Ia) fibroblasts. Pediatr Res. 2002 Nov;52(5):645-51. | | 6. von Nicolai H, Esser P, Lauer E: Partial purification and properties of neuraminidase from Bifidobacterium lactentis. Hoppe Seylers Z Physiol Chem. 1981 Feb;362(2):153-62. | | 7. Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. | | 8. Rodriguez-Aparicio LB, Ferrero MA, Reglero A: N-acetyl-D-neuraminic acid synthesis in Escherichia coli K1 occurs through condensation of N-acetyl-D-mannosamine and pyruvate. Biochem J. 1995 Jun 1;308 ( Pt 2):501-5. | | 9. https://www.ncbi.nlm.nih.gov/pubmed/?term=15280054 | | 10. https://www.ncbi.nlm.nih.gov/pubmed/?term=15936003 | | 11. https://www.ncbi.nlm.nih.gov/pubmed/?term=8154046 |
|
|---|