Identification Common Name Tauroursodeoxycholic acid Class Small Molecule Description Tauroursodeoxycholic acid is a bile acid also known as TUDCA formed in the liver by conjugation of deoxycholate with taurine, usually as the sodium salt. TUDCA is able to prevent apoptosis and protect mitochondria from cellular elements that would otherwise interfere with energy production. One of these elements is a protein called Bax. TUDCA plays an important role in preventing Bax from being transported to the mitochondria. Bile acids are steroid acids found predominantly in the bile of mammals. The distinction between different bile acids is minute, depending only on the presence or absence of hydroxyl groups on positions 3, 7, and 12. Bile acids are physiological detergents that facilitate excretion, absorption, and transport of fats and sterols in the intestine and liver. Bile acids are also steroidal amphipathic molecules derived from the catabolism of cholesterol. They modulate bile flow and lipid secretion, are essential for the absorption of dietary fats and vitamins, and have been implicated in the regulation of all the key enzymes involved in cholesterol homeostasis. Bile acids recirculate through the liver, bile ducts, small intestine and portal vein to form an enterohepatic circuit. They exist as anions at physiological pH and, consequently, require a carrier for transport across the membranes of the enterohepatic tissues. The unique detergent properties of bile acids are essential for the digestion and intestinal absorption of hydrophobic nutrients. Bile acids have potent toxic properties (e.g. membrane disruption) and there are a plethora of mechanisms to limit their accumulation in blood and tissues (PMID: 11316487, 16037564, 12576301, 11907135). Contaminant Sources FooDB Chemicals
HMDB Contaminants - Feces Contaminant Type Not Available Chemical Structure Synonyms Value Source Tauroursodeoxycholate Generator 2-(((3-alpha,5-beta,7-beta)-3,7-Dihydroxy-24-oxocholan-24-yl) amino)ethanesulfonate HMDB 2-(((3-alpha,5-beta,7-beta)-3,7-Dihydroxy-24-oxocholan-24-yl) amino)ethanesulfonic acid HMDB 2-(((3-alpha,5-beta,7-beta)-3,7-Dihydroxy-24-oxocholan-24-yl)amino)-ethanesulfonate HMDB 2-(((3-alpha,5-beta,7-beta)-3,7-Dihydroxy-24-oxocholan-24-yl)amino)-ethanesulfonic acid HMDB 3a,7b-Dihydroxy-5b-cholanoyltaurine HMDB N-(3-alpha,7-beta-Dihydroxy-5-beta-cholan-24-oyl)-taurine HMDB TUDCA HMDB UR 906 HMDB Ursodeoxycholyltaurine HMDB Tauroursodeoxycholic acid, monosodium salt, (3alpha,5beta,7alpha)-isomer MeSH, HMDB Tauroursodeoxycholic acid, (3alpha,5alpha,7alpha)-isomer MeSH, HMDB Tauroursodeoxycholic acid, monosodium salt, (3alpha,7alpha)-isomer MeSH, HMDB (4R)-4-[(1S,2S,5R,9S,10R,11S,14R,15R)-5,9-Dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-N-(2-sulfoethyl)pentanimidate Generator, HMDB (4R)-4-[(1S,2S,5R,9S,10R,11S,14R,15R)-5,9-Dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-N-(2-sulphoethyl)pentanimidate Generator, HMDB (4R)-4-[(1S,2S,5R,9S,10R,11S,14R,15R)-5,9-Dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-N-(2-sulphoethyl)pentanimidic acid Generator, HMDB
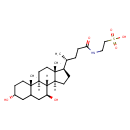
Chemical Formula C26 H45 NO6 S Average Molecular Mass 499.704 g/mol Monoisotopic Mass 499.297 g/mol CAS Registry Number 14605-22-2 IUPAC Name 2-[(4R)-4-[(1S,2S,5R,9S,10R,11S,14R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanamido]ethane-1-sulfonic acid Traditional Name 2-[(4R)-4-[(1S,2S,5R,9S,10R,11S,14R,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanamido]ethanesulfonic acid SMILES [H][C@@]12CC[C@H]([C@H](C)CCC(=O)NCCS(O)(=O)=O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](O)CC2C[C@H](O)CC[C@]12C InChI Identifier InChI=1S/C26H45NO6S/c1-16(4-7-23(30)27-12-13-34(31,32)33)19-5-6-20-24-21(9-11-26(19,20)3)25(2)10-8-18(28)14-17(25)15-22(24)29/h16-22,24,28-29H,4-15H2,1-3H3,(H,27,30)(H,31,32,33)/t16-,17?,18-,19-,20+,21+,22+,24+,25+,26-/m1/s1 InChI Key BHTRKEVKTKCXOH-VSHSPWMTSA-N Spectra Spectra Spectrum Type Description Splash Key View Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive splash10-053r-0212900000-14bc097af3f7499753d7 Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive splash10-004i-4214259000-4a72734ef61b01c2f556 Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_2_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_2_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_2_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_2_5) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_2_6) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_2_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_2_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_2_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_2_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_2_5) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_2_6) - 70eV, Positive Not Available Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-00e9-0801920000-ecfc97935416325b3ca5 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-00di-2901100000-9a3cf7f2963c041be363 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-006x-8904100000-98cfa2606dbba4ef47a0 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-000t-2001900000-4adf0e40de3812e8d0ba Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-001i-6404900000-4d05b6f0352aebf4228d Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-001l-9102000000-63892e6e06f1123f8bfc Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-0002-0000900000-31f17d1d48b7afdf3bae Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-0002-0000900000-f4f03ef481870394f9e2 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-001i-9100300000-654e1207ef97c7662b6e Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-0gx0-0000940000-9827b7f4a5b2f54a7bd4 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-0nmi-4639730000-a8491d87ed7e9a353ee5 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-0f7x-3893000000-cc5d0b26da084a4d3a35 Spectrum