| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:50:03 UTC |
|---|
| Update Date | 2016-11-09 01:17:34 UTC |
|---|
| Accession Number | CHEM022733 |
|---|
| Identification |
|---|
| Common Name | 2-hydroxyclomipramine glucuronide |
|---|
| Class | Small Molecule |
|---|
| Description | 2-Amino-3-methylbenzoate is a metabolite of lidocaine. Lidocaine, Xylocaine, or lignocaine is a common local anesthetic and antiarrhythmic drug. Lidocaine is used topically to relieve itching, burning and pain from skin inflammations, injected as a dental anesthetic or as a local anesthetic for minor surgery. (Wikipedia) |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
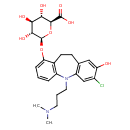
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Amino-3-methylbenzoic acid | Generator | | 2-Amino-3-methylbenzoic acid, monopotassium salt | HMDB |
|
|---|
| Chemical Formula | C25H31ClN2O8 |
|---|
| Average Molecular Mass | 522.975 g/mol |
|---|
| Monoisotopic Mass | 522.177 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2S,3S,4S,5R,6S)-6-({14-chloro-2-[3-(dimethylamino)propyl]-13-hydroxy-2-azatricyclo[9.4.0.0³,⁸]pentadeca-1(11),3(8),4,6,12,14-hexaen-7-yl}oxy)-3,4,5-trihydroxyoxane-2-carboxylic acid |
|---|
| Traditional Name | (2S,3S,4S,5R,6S)-6-({14-chloro-2-[3-(dimethylamino)propyl]-13-hydroxy-2-azatricyclo[9.4.0.0³,⁸]pentadeca-1(11),3(8),4,6,12,14-hexaen-7-yl}oxy)-3,4,5-trihydroxyoxane-2-carboxylic acid |
|---|
| SMILES | CN(C)CCCN1C2=C(CCC3=C1C=C(Cl)C(O)=C3)C(O[C@@H]1O[C@@H]([C@@H](O)[C@H](O)[C@H]1O)C(O)=O)=CC=C2 |
|---|
| InChI Identifier | InChI=1S/C25H31ClN2O8/c1-27(2)9-4-10-28-16-5-3-6-19(14(16)8-7-13-11-18(29)15(26)12-17(13)28)35-25-22(32)20(30)21(31)23(36-25)24(33)34/h3,5-6,11-12,20-23,25,29-32H,4,7-10H2,1-2H3,(H,33,34)/t20-,21-,22+,23-,25+/m0/s1 |
|---|
| InChI Key | FHDAIUXAPWZOKI-LYVDORBWSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as aminobenzoic acids. These are benzoic acids containing an amine group attached to the benzene moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
| Direct Parent | Aminobenzoic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aminobenzoic acid
- Benzoic acid
- Benzoyl
- Aminotoluene
- Aniline or substituted anilines
- Toluene
- Vinylogous amide
- Amino acid or derivatives
- Amino acid
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Amine
- Organonitrogen compound
- Organooxygen compound
- Primary amine
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4l-9200210000-cd56be6b64bbd849cc1a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00fs-2307490000-9e78a5c084cfc254449d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-1219210000-48b6eddd0e13b3110727 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-06r2-4159000000-5edaefc0e37f18468db6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-006t-1009160000-22cf2bddfad44aef1079 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000t-1019000000-2bc2ed2ba777154d56b9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-9355000000-b1377706a78b778b87ef | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0060680 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 78101 |
|---|
| Kegg Compound ID | C16571 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|