| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:42:39 UTC |
|---|
| Update Date | 2026-03-31 18:33:32 UTC |
|---|
| Accession Number | CHEM022623 |
|---|
| Identification |
|---|
| Common Name | 8-Hydroxyamoxapine |
|---|
| Class | Small Molecule |
|---|
| Description | 8-Hydroxyamoxapine is a metabolite of amoxapine. Amoxapine (Amokisan, Asendin, Asendis, Defanyl, Demolox, Moxadil) is a tetracyclic antidepressant of the dibenzoxazepine family, though it is often classified as a secondary amine tricyclic antidepressant. It is the N-demethylated metabolite of Loxapine. (Wikipedia) |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
- STOFF IDENT Compounds
|
|---|
| Contaminant Type | Not Available |
|---|
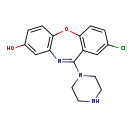
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C17H16ClN3O2 |
|---|
| Average Molecular Mass | 329.781 g/mol |
|---|
| Monoisotopic Mass | 329.093 g/mol |
|---|
| CAS Registry Number | 61443-78-5 |
|---|
| IUPAC Name | 13-chloro-10-(piperazin-1-yl)-2-oxa-9-azatricyclo[9.4.0.0³,⁸]pentadeca-1(11),3(8),4,6,9,12,14-heptaen-6-ol |
|---|
| Traditional Name | 13-chloro-10-(piperazin-1-yl)-2-oxa-9-azatricyclo[9.4.0.0³,⁸]pentadeca-1(11),3(8),4,6,9,12,14-heptaen-6-ol |
|---|
| SMILES | OC1=CC2=C(OC3=C(C=C(Cl)C=C3)C(=N2)N2CCNCC2)C=C1 |
|---|
| InChI Identifier | InChI=1S/C17H16ClN3O2/c18-11-1-3-15-13(9-11)17(21-7-5-19-6-8-21)20-14-10-12(22)2-4-16(14)23-15/h1-4,9-10,19,22H,5-8H2 |
|---|
| InChI Key | QDWNOKXUZTYVGO-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as dibenzoxazepines. Dibenzoxazepines are compounds containing a dibenzoxazepine moiety, which consists of two benzene connected by an oxazepine ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzoxazepines |
|---|
| Sub Class | Dibenzoxazepines |
|---|
| Direct Parent | Dibenzoxazepines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Dibenzoxazepine
- Diaryl ether
- 1-hydroxy-2-unsubstituted benzenoid
- Aryl chloride
- Aryl halide
- 1,4-diazinane
- Piperazine
- Imidolactam
- Benzenoid
- Amidine
- Carboxylic acid amidine
- Secondary aliphatic amine
- Oxacycle
- Ether
- Azacycle
- Secondary amine
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Organic nitrogen compound
- Organic oxygen compound
- Organohalogen compound
- Organochloride
- Organonitrogen compound
- Organooxygen compound
- Amine
- Organopnictogen compound
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0f9f-9182000000-b8b8e53cfb28088f2cfd | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0079-9107000000-22028c9ad311da80bb01 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0009000000-5e07284fce8eb5819c25 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0029000000-25daabc66b1e281ec7ab | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0cdr-6970000000-72155ae7835c0318d6ec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0009000000-b5b8162a95df4f9068f7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-0039000000-5114673474a52e69ca5a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9140000000-9d2aee81881a32ddfd54 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0009000000-317a80daeb1f485525d0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-1019000000-03dbb1ddc44bd4b23e2f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001i-2092000000-c2da73db1263ba71f06e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0009000000-2a0b3ee37f0986f8fe94 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0039000000-dfb26f6afe4e3d81cb24 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-1391000000-8ef4a881ac95f66f665c | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0060801 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | 8-Hydroxyamoxapine |
|---|
| Chemspider ID | 39784 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 43656 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|