| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:42:29 UTC |
|---|
| Update Date | 2016-11-09 01:17:33 UTC |
|---|
| Accession Number | CHEM022621 |
|---|
| Identification |
|---|
| Common Name | 5'-Hydroxymethyl meloxicam |
|---|
| Class | Small Molecule |
|---|
| Description | 5'-Hydroxymethyl meloxicam is a metabolite of meloxicam. Meloxicam is a nonsteroidal anti-inflammatory drug (NSAID) with analgesic and fever reducer effects. It is a derivative of oxicam, closely related to piroxicam, and falls in the enolic acid group of NSAIDs. It was developed by Boehringer-Ingelheim. (Wikipedia) |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
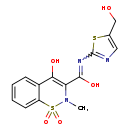
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 6-Thioguanosine-5'- O- triphosphoric acid | HMDB |
|
|---|
| Chemical Formula | C14H13N3O5S2 |
|---|
| Average Molecular Mass | 367.400 g/mol |
|---|
| Monoisotopic Mass | 367.030 g/mol |
|---|
| CAS Registry Number | 130262-92-9 |
|---|
| IUPAC Name | 4-hydroxy-N-[5-(hydroxymethyl)-1,3-thiazol-2-yl]-2-methyl-1,1-dioxo-2H-1λ⁶,2-benzothiazine-3-carboximidic acid |
|---|
| Traditional Name | 4-hydroxy-N-[5-(hydroxymethyl)-1,3-thiazol-2-yl]-2-methyl-1,1-dioxo-1λ⁶,2-benzothiazine-3-carboximidic acid |
|---|
| SMILES | CN1C(C(O)=NC2=NC=C(CO)S2)=C(O)C2=CC=CC=C2S1(=O)=O |
|---|
| InChI Identifier | InChI=1S/C14H13N3O5S2/c1-17-11(13(20)16-14-15-6-8(7-18)23-14)12(19)9-4-2-3-5-10(9)24(17,21)22/h2-6,18-19H,7H2,1H3,(H,15,16,20) |
|---|
| InChI Key | LOOSAJMIEADILV-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzothiazines. These are organic compounds containing a benzene fused to a thiazine ring (a six-membered ring with four carbon atoms, one nitrogen atom and one sulfur atom). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzothiazines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzothiazines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzothiazine
- 2,5-disubstituted 1,3-thiazole
- Ortho-thiazine
- Organosulfonic acid amide
- Benzenoid
- Azole
- Organic sulfonic acid or derivatives
- Heteroaromatic compound
- Organosulfonic acid or derivatives
- Thiazole
- Carboximidic acid
- Carboximidic acid derivative
- Azacycle
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Primary alcohol
- Organooxygen compound
- Organonitrogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Organic oxide
- Hydrocarbon derivative
- Alcohol
- Organopnictogen compound
- Aromatic alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0gdi-2924000000-1f64709c73fb12ef4ac2 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-00kf-4496670000-ae8b75378286073b9896 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0v00-0609000000-c0786eed07ad26d3549e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-1910000000-655173c0863f6bd6f41c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01t9-3900000000-cfde0b98ad34fd7b002b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014r-1239000000-70eb7d01544ed934dea6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00kr-2359000000-b6c14388f58149caba6a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-08i0-9810000000-46051c4bb4f1cb44e3ea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0019000000-b782035a0db13e8d9dff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03xr-0925000000-6d5be46b88df993a673a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03di-1910000000-8cd2a49a0883d3f8c7d7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0gb9-0309000000-a1da1420ccfa1c78d8b9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03dl-5930000000-06629d4204c8186fcdd0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03xr-6963000000-e22cf9e39834088cd601 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0060780 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 21896764 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 131769948 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|