| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:42:16 UTC |
|---|
| Update Date | 2016-11-09 01:17:32 UTC |
|---|
| Accession Number | CHEM022617 |
|---|
| Identification |
|---|
| Common Name | 4-Carboxynevirapine |
|---|
| Class | Small Molecule |
|---|
| Description | 4-Carboxynevirapine is a metabolite of nevirapine. Nevirapine, also marketed under the trade name Viramune, is a non-nucleoside reverse transcriptase inhibitor (NNRTI) used to treat HIV-1 infection and AIDS. As with other antiretroviral drugs, HIV rapidly develops resistance if nevirapine is used alone, so recommended therapy consists of combinations of three or more antiretrovirals. (Wikipedia) |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3'-Azido-3'-deoxy-5'- O-b-D-glucopyranuronosylthymidine | HMDB | | 3'-Azido-3'-deoxy-5'- O-β-D-glucopyranuronosylthymidine | HMDB | | AZT glucuronide | HMDB | | 3'-Azido-3'-deoxy-5'-O-beta-glucopyranuronosylthymidine | HMDB | | GAZT | HMDB | | AZT-5'-glucuronide | HMDB |
|
|---|
| Chemical Formula | C15H12N4O3 |
|---|
| Average Molecular Mass | 296.281 g/mol |
|---|
| Monoisotopic Mass | 296.091 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
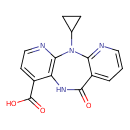
| IUPAC Name | 2-cyclopropyl-10-oxo-2,4,9,15-tetraazatricyclo[9.4.0.0³,⁸]pentadeca-1(15),3(8),4,6,11,13-hexaene-7-carboxylic acid |
|---|
| Traditional Name | 2-cyclopropyl-10-oxo-2,4,9,15-tetraazatricyclo[9.4.0.0³,⁸]pentadeca-1(15),3(8),4,6,11,13-hexaene-7-carboxylic acid |
|---|
| SMILES | OC(=O)C1=CC=NC2=C1NC(=O)C1=CC=CN=C1N2C1CC1 |
|---|
| InChI Identifier | InChI=1S/C15H12N4O3/c20-14-10-2-1-6-16-12(10)19(8-3-4-8)13-11(18-14)9(15(21)22)5-7-17-13/h1-2,5-8H,3-4H2,(H,18,20)(H,21,22) |
|---|
| InChI Key | WDXCMIDQWGYHIN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alkyldiarylamines. These are tertiary alkylarylamines having two aryl and one alkyl groups attached to the amino group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic nitrogen compounds |
|---|
| Class | Organonitrogen compounds |
|---|
| Sub Class | Amines |
|---|
| Direct Parent | Alkyldiarylamines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alkyldiarylamine
- Pyrido-para-diazepine
- Pyridine carboxylic acid
- Pyridine carboxylic acid or derivatives
- Para-diazepine
- Pyridine
- Imidolactam
- Heteroaromatic compound
- Vinylogous amide
- Carboxamide group
- Lactam
- Secondary carboxylic acid amide
- Azacycle
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organic oxide
- Organooxygen compound
- Organic oxygen compound
- Organopnictogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0kuu-0490000000-868c3fa487d18d561bcf | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0fn9-8497000000-b1059cc66c8651a2f9f6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0090000000-03fc360d0dd52eeaf3eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ufs-0090000000-b9d8e1529284c8d08e6a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gvx-8190000000-8eeb53c98632b1d30dad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udj-0090000000-3758c78030929ebc316c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0090000000-b0d5580a8d1a49c7fa29 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bt9-0490000000-e08953300981317eca97 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-002b-0090000000-ab5963bf94cd943f310b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0090000000-e68a8916533f9476ac08 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03dr-0290000000-28294370a3a15f4e979c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0090000000-2d8d283e12d342c2f5a3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0zfr-0090000000-ee1b50340d9632c80d4e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bt9-0190000000-b405eca4e4edb6268eb9 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0060759 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 8884376 |
|---|
| ChEBI ID | 169675 |
|---|
| PubChem Compound ID | 10709035 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|