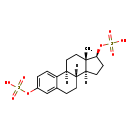Identification Common Name 17-Beta-Estradiol-3,17-beta-sulfate Class Small Molecule Description 17-Beta-Estradiol-3,17-beta-sulfate is found in milk. Contaminant Sources FooDB Chemicals
HMDB Contaminants - Urine Contaminant Type Not Available Chemical Structure Synonyms Value Source 17-b-Estradiol-3,17-b-sulfate Generator 17-b-Estradiol-3,17-b-sulfuric acid Generator 17-b-Estradiol-3,17-b-sulphate Generator 17-b-Estradiol-3,17-b-sulphuric acid Generator 17-beta-Estradiol-3,17-beta-sulfuric acid Generator 17-beta-Estradiol-3,17-beta-sulphate Generator 17-beta-Estradiol-3,17-beta-sulphuric acid Generator 17-Β-estradiol-3,17-β-sulfate Generator 17-Β-estradiol-3,17-β-sulfuric acid Generator 17-Β-estradiol-3,17-β-sulphate Generator 17-Β-estradiol-3,17-β-sulphuric acid Generator 17beta-Estradiol-3,17-disulfate MeSH Estradiol 3,17-disulfate, (17alpha)-isomer MeSH Estradiol 3,17-disulfate, dipotassium salt, (17beta)-isomer MeSH Estradiol 3,17-disulfate MeSH Estradiol 3,17-disulfate, disodium salt, (17beta)-isomer MeSH e2-3S-17S HMDB
Chemical Formula C18 H24 O8 S2 Average Molecular Mass 432.508 g/mol Monoisotopic Mass 432.091 g/mol CAS Registry Number Not Available IUPAC Name [(1S,10R,11S,14S,15S)-15-methyl-5-(sulfooxy)tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadeca-2,4,6-trien-14-yl]oxidanesulfonic acid Traditional Name [(1S,10R,11S,14S,15S)-15-methyl-5-(sulfooxy)tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadeca-2,4,6-trien-14-yl]oxidanesulfonic acid SMILES [H][C@@]12CC[C@H](OS(O)(=O)=O)[C@@]1(C)CC[C@]1([H])C3=CC=C(OS(O)(=O)=O)C=C3CC[C@@]21[H] InChI Identifier InChI=1S/C18H24O8S2/c1-18-9-8-14-13-5-3-12(25-27(19,20)21)10-11(13)2-4-15(14)16(18)6-7-17(18)26-28(22,23)24/h3,5,10,14-17H,2,4,6-9H2,1H3,(H,19,20,21)(H,22,23,24)/t14-,15-,16+,17+,18+/m1/s1 InChI Key VPLAJGAMHNQZIY-ZBRFXRBCSA-N