| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:39:25 UTC |
|---|
| Update Date | 2016-11-09 01:17:32 UTC |
|---|
| Accession Number | CHEM022556 |
|---|
| Identification |
|---|
| Common Name | 2-Hydroxyestrone sulfate |
|---|
| Class | Small Molecule |
|---|
| Description | 2-Hydroxyestrone sulfate is a sulfate derivative of Estrone. Estrone (also oestrone) is an estrogenic hormone secreted by the ovary. Its molecular formula is C18H22O2. estrone has a melting point of 254.5 degrees Celsius. estrone is one of the three estrogens, which also include estriol and estradiol. estrone is the least prevalent of the three hormones, estradiol being prevalent almost always in a female body, estriol being prevalent primarily during pregnancy. estrone sulfate is relevant to health and disease due to its conversion to estrone sulfate, a long-lived derivative of estrone. estrone sulfate acts as a pool of estrone which can be converted as needed to the more active estradiol. |
|---|
| Contaminant Sources | - FooDB Chemicals
- HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
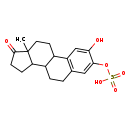
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Hydroxyestrone sulfuric acid | Generator | | 2-Hydroxyestrone sulphate | Generator | | 2-Hydroxyestrone sulphuric acid | Generator | | 1,3,5(10)-Triene-2,3-diol-17-one 3-sulfate | HMDB | | 1,3,5(10)-Triene-2,3-diol-17-one 3-sulphate | HMDB | | 2,3-Dihydroxyestra-1,3,5(10)-trien-17-one 3-sulfate | HMDB | | 2,3-Dihydroxyestra-1,3,5(10)-trien-17-one 3-sulphate | HMDB | | 2HEn-3S | HMDB | | Catecholestrogen sulfate | HMDB | | Catecholestrogen sulphate | HMDB | | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadeca-2,4,6-trien-5-yl}oxidanesulfonate | HMDB | | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadeca-2,4,6-trien-5-yl}oxidanesulphonate | HMDB | | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadeca-2,4,6-trien-5-yl}oxidanesulphonic acid | HMDB | | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0,.0,]heptadeca-2,4,6-trien-5-yl}oxidanesulfonate | HMDB | | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0,.0,]heptadeca-2,4,6-trien-5-yl}oxidanesulphonate | HMDB | | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0,.0,]heptadeca-2,4,6-trien-5-yl}oxidanesulphonic acid | HMDB |
|
|---|
| Chemical Formula | C18H21O6S |
|---|
| Average Molecular Mass | 365.421 g/mol |
|---|
| Monoisotopic Mass | 365.106 g/mol |
|---|
| CAS Registry Number | 1240-04-6 |
|---|
| IUPAC Name | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-2(7),3,5-trien-5-yl}oxidanesulfonic acid |
|---|
| Traditional Name | {4-hydroxy-15-methyl-14-oxotetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-2(7),3,5-trien-5-yl}oxidanesulfonic acid |
|---|
| SMILES | C[C@]12CCC3C(CCC4=C3C=C(O)C(OS([O-])(=O)=O)=C4)C1CCC2=O |
|---|
| InChI Identifier | InChI=1S/C18H22O6S/c1-18-7-6-11-12(14(18)4-5-17(18)20)3-2-10-8-16(24-25(21,22)23)15(19)9-13(10)11/h8-9,11-12,14,19H,2-7H2,1H3,(H,21,22,23)/p-1/t11?,12?,14?,18-/m0/s1 |
|---|
| InChI Key | DTMWSKSAMRZGQH-PEDDLLMLSA-M |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as sulfated steroids. These are sterol lipids containing a sulfate group attached to the steroid skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Sulfated steroids |
|---|
| Direct Parent | Sulfated steroids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Sulfated steroid skeleton
- Estrane-skeleton
- 2-hydroxysteroid
- Hydroxysteroid
- 17-oxosteroid
- Oxosteroid
- Phenanthrene
- Arylsulfate
- Tetralin
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Benzenoid
- Sulfuric acid ester
- Sulfate-ester
- Sulfuric acid monoester
- Organic sulfuric acid or derivatives
- Ketone
- Organooxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organic oxygen compound
- Organic oxide
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0019000000-502ef23ca1af4c129c95 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014s-1369000000-3d0567be3e828fd1435b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0umm-3396000000-6870ba253a121813102d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-0cec9790eaa0e659a484 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00kr-0093000000-acfc5095321f966c1f43 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05o9-5090000000-64e770a5900ee5227323 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-f9549c8f91b5c3bae9e2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014j-2009000000-8994753ce191ba676aa7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01ot-7093000000-95ce709641b15796d0a1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0019000000-39f2458cfa21660c2105 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-1093000000-7707105d2fc84e4eebfb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-06r2-4795000000-845c908a83df4bc73f98 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0012622 |
|---|
| FooDB ID | FDB029153 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 11467601 |
|---|
| ChEBI ID | 175686 |
|---|
| PubChem Compound ID | 22569148 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|