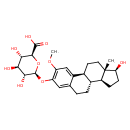| (17beta)-17-Hydroxy-2-methoxyestra-1,3,5(10)-trien-3-yl beta-D-glucopyranosiduronic acid | ChEBI |
| 2-MeOE2 3g | ChEBI |
| 2-Methoxy-17beta-estradiol 3-beta-D-glucuronide | ChEBI |
| 2-Methoxy-17beta-estradiol 3-beta-glucuronide | ChEBI |
| 2-Methoxy-17beta-estradiol 3-glucosiduronic acid | ChEBI |
| 2-Methoxy-17beta-estradiol 3-glucuronide | ChEBI |
| 2-Methoxy-17beta-estradiol 3-O-(beta-D-glucuronic acid) | ChEBI |
| 2-Methoxy-17beta-estradiol 3-O-glucuronide | ChEBI |
| 2-Methoxy-estradiol-17beta 3-glucuronide | ChEBI |
| 2-Methoxyestradiol 3-glucuronide | ChEBI |
| (17b)-17-Hydroxy-2-methoxyestra-1,3,5(10)-trien-3-yl b-D-glucopyranosiduronate | Generator |
| (17b)-17-Hydroxy-2-methoxyestra-1,3,5(10)-trien-3-yl b-D-glucopyranosiduronic acid | Generator |
| (17beta)-17-Hydroxy-2-methoxyestra-1,3,5(10)-trien-3-yl beta-D-glucopyranosiduronate | Generator |
| (17Β)-17-hydroxy-2-methoxyestra-1,3,5(10)-trien-3-yl β-D-glucopyranosiduronate | Generator |
| (17Β)-17-hydroxy-2-methoxyestra-1,3,5(10)-trien-3-yl β-D-glucopyranosiduronic acid | Generator |
| 2-Methoxy-17b-estradiol 3-b-D-glucuronide | Generator |
| 2-Methoxy-17β-estradiol 3-β-D-glucuronide | Generator |
| 2-Methoxy-17b-estradiol 3-b-glucuronide | Generator |
| 2-Methoxy-17β-estradiol 3-β-glucuronide | Generator |
| 2-Methoxy-17b-estradiol 3-glucosiduronate | Generator |
| 2-Methoxy-17b-estradiol 3-glucosiduronic acid | Generator |
| 2-Methoxy-17beta-estradiol 3-glucosiduronate | Generator |
| 2-Methoxy-17β-estradiol 3-glucosiduronate | Generator |
| 2-Methoxy-17β-estradiol 3-glucosiduronic acid | Generator |
| 2-Methoxy-17b-estradiol 3-glucuronide | Generator |
| 2-Methoxy-17β-estradiol 3-glucuronide | Generator |
| 2-Methoxy-17b-estradiol 3-O-(b-D-glucuronate) | Generator |
| 2-Methoxy-17b-estradiol 3-O-(b-D-glucuronic acid) | Generator |
| 2-Methoxy-17beta-estradiol 3-O-(beta-D-glucuronate) | Generator |
| 2-Methoxy-17β-estradiol 3-O-(β-D-glucuronate) | Generator |
| 2-Methoxy-17β-estradiol 3-O-(β-D-glucuronic acid) | Generator |
| 2-Methoxy-17b-estradiol 3-O-glucuronide | Generator |
| 2-Methoxy-17β-estradiol 3-O-glucuronide | Generator |
| 2-Methoxy-estradiol-17β 3-glucuronide | Generator |
| 2-Methoxy-estradiol-17b 3-glucuronide | Generator |