| Synonyms | | Value | Source |
|---|
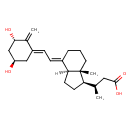
| Calcitroate | Generator | | (3R)-3-[(1R,3AR,4E,7ar)- 4-[(2Z)-2-[(3R,5R)-3,5- dihydroxy-2-methylene-cyclohexylidene]ethylidene] -7a-methyl-2,3,3a,5,6,7-hexahydro-1H -inden-1-yl]butanoate | HMDB | | (3R)-3-[(1R,3AR,4E,7ar)- 4-[(2Z)-2-[(3R,5R)-3,5- dihydroxy-2-methylene-cyclohexylidene]ethylidene] -7a-methyl-2,3,3a,5,6,7-hexahydro-1H -inden-1-yl]butanoic acid | HMDB | | (3R)-3-[(1R,3AS,4E,7ar)-4-[(2Z)-2-[(3R,5S)-3,5-dihydroxy-2 methylidenecyclohexylidene]ethylidene]-7a-methyl-2,3,3a,5,6,7-hexahydro-1H-inden-1-yl]butanoate | HMDB | | (3R)-3-[(1R,3AS,4E,7ar)-4-[(2Z)-2-[(3R,5S)-3,5-dihydroxy-2 methylidenecyclohexylidene]ethylidene]-7a-methyl-2,3,3a,5,6,7-hexahydro-1H-inden-1-yl]butanoic acid | HMDB | | 1 alpha-Hydroxy-23 carboxy-24,25,26,27-tetranorvitamin D(3) | HMDB | | 1a-Hydroxycalcioate | HMDB | | 1a-Hydroxycalcioic acid | HMDB | | 1alpha,3beta-Dihydroxy-24-nor-9,10-seco-5,7,10(19)-cholatrien-23-Oate | HMDB | | 1alpha,3beta-Dihydroxy-24-nor-9,10-seco-5,7,10(19)-cholatrien-23-Oic acid | HMDB | | 1alpha-Hydroxy-23-carboxytetranorvitamin D | HMDB | | Calcitroic acid (D3) | HMDB | | 1 alpha,3 beta-Dihydroxy-24-nor-9,10- seco-5,7,10(19)-cholatrien-23-Oic acid | HMDB | | 1 alpha-Hydroxy-23-carboxytetranorvitamin D | HMDB | | (3R)-3-[(1R,3AS,4E,7ar)-4-{2-[(1E,3S,5R)-3,5-dihydroxy-2-methylidenecyclohexylidene]ethylidene}-7a-methyl-octahydro-1H-inden-1-yl]butanoate | HMDB | | Calcitroic acid | HMDB |
|
|---|
| InChI Identifier | InChI=1S/C23H34O4/c1-14(11-22(26)27)19-8-9-20-16(5-4-10-23(19,20)3)6-7-17-12-18(24)13-21(25)15(17)2/h6-7,14,18-21,24-25H,2,4-5,8-13H2,1,3H3,(H,26,27)/b16-6+,17-7+/t14-,18-,19-,20+,21+,23-/m1/s1 |
|---|