| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:30:01 UTC |
|---|
| Update Date | 2026-03-26 19:34:47 UTC |
|---|
| Accession Number | CHEM022364 |
|---|
| Identification |
|---|
| Common Name | Drotaverine |
|---|
| Class | Small Molecule |
|---|
| Description | Drotaverine (INN, also known as drotaverin) is an antispasmodic drug, structurally related to papaverine. Drotaverine is a selective inhibitor of phosphodiesterase 4, and has no anticholinergic effects. Drotaverine has been shown to possess dose-dependant analgesic effects in animal models. One small study has shown drotaverine to be eliminated mainly non-renally. |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
- STOFF IDENT Compounds
|
|---|
| Contaminant Type | Not Available |
|---|
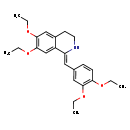
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Drotin | Kegg | | Drotaverin | HMDB | | Nospan | HMDB | | Isodihydroperparine | HMDB | | NO-Shpa | HMDB | | Dihydroisoperparine | HMDB | | Drotaverin hydrochloride | HMDB | | NO-Spa | HMDB | | Nospanum | HMDB |
|
|---|
| Chemical Formula | C24H31NO4 |
|---|
| Average Molecular Mass | 397.507 g/mol |
|---|
| Monoisotopic Mass | 397.225 g/mol |
|---|
| CAS Registry Number | 985-12-6 |
|---|
| IUPAC Name | (1Z)-1-[(3,4-diethoxyphenyl)methylidene]-6,7-diethoxy-1,2,3,4-tetrahydroisoquinoline |
|---|
| Traditional Name | drotaverine |
|---|
| SMILES | CCOC1=C(OCC)C=C(\C=C2/NCCC3=CC(OCC)=C(OCC)C=C23)C=C1 |
|---|
| InChI Identifier | InChI=1S/C24H31NO4/c1-5-26-21-10-9-17(14-22(21)27-6-2)13-20-19-16-24(29-8-4)23(28-7-3)15-18(19)11-12-25-20/h9-10,13-16,25H,5-8,11-12H2,1-4H3/b20-13- |
|---|
| InChI Key | OMFNSKIUKYOYRG-MOSHPQCFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as tetrahydroisoquinolines. These are tetrahydrogenated isoquinoline derivatives. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Tetrahydroisoquinolines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Tetrahydroisoquinolines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tetrahydroisoquinoline
- Phenoxy compound
- Phenol ether
- Alkyl aryl ether
- Aralkylamine
- Monocyclic benzene moiety
- Benzenoid
- Secondary aliphatic amine
- Enamine
- Ether
- Secondary amine
- Azacycle
- Organonitrogen compound
- Amine
- Organooxygen compound
- Organopnictogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-01b9-0029000000-cc21574b335ef828553c | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-006t-0109000000-7cad269f41adee8417ce | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00vm-0938000000-6d87baa6b9b9001f72b3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fai-0903000000-32461a4dd8213a4ccaae | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00kb-0009000000-ab1aeeb006841b3fa9c8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01b9-0009000000-06822dcfe079230d41a9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01pc-0019000000-acda050fb3e73672fc3e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0009000000-8c6d856694872eafbde7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-006t-0009000000-753a599270e1789b1d24 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-006x-0169000000-2f0a9da1a5938c47a4e9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0009000000-a71d93a03163a6601571 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0009000000-c70358ec5190b857e386 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00du-0019000000-7c1afa91b9c7710b0f53 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB06751 |
|---|
| HMDB ID | HMDB0015669 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Drotaverine |
|---|
| Chemspider ID | 1361582 |
|---|
| ChEBI ID | 665387 |
|---|
| PubChem Compound ID | 1712095 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|