| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:21:42 UTC |
|---|
| Update Date | 2016-11-09 01:17:26 UTC |
|---|
| Accession Number | CHEM022149 |
|---|
| Identification |
|---|
| Common Name | Bacitracin |
|---|
| Class | Small Molecule |
|---|
| Description | Bacitracin is a combination of at least 9 bacitracins. 60-80% of commercially prepared bacitracin is bacitracin A. The bacillus that produces bacitracin was first isolated from a knee scrape in 1945 from the knee wound of a child named Margaret Tracy.
Bacitracin was granted FDA approval on 29 July 1948. |
|---|
| Contaminant Sources | - FooDB Chemicals
- HMDB Contaminants - Urine
- STOFF IDENT Compounds
|
|---|
| Contaminant Type | Not Available |
|---|
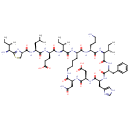
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Bacitracin a2a | ChEBI | | A.L. labs brand OF bacitracin zinc salt | HMDB | | Ak tracin | HMDB | | Akorn brand OF bacitracin | HMDB | | Lee brand OF bacitracin | HMDB | | Pharmascience brand OF bacitracin | HMDB | | Reed and carnrick brand OF bacitracin | HMDB | | Topitracin | HMDB | | Zinc bacitracin | HMDB | | Bacitracin a | HMDB | | Baci-RX | HMDB | | Bacitracin pharmascience brand | HMDB | | Bacitracin zinc complex | HMDB | | Bacitracine martinet | HMDB | | McNeil brand OF bacitracin | HMDB | | OcuTracin | HMDB | | Baci RX | HMDB | | Bacitracin akorn brand | HMDB | | Bacitracin mcneil brand | HMDB | | Bacitracin zinc | HMDB | | Martinet, bacitracine | HMDB | | Ocu tracin | HMDB | | Ocu-tracin | HMDB | | Ocumed brand OF bacitracin | HMDB | | Zeba RX | HMDB | | Zeba-RX | HMDB | | Zinc, bacitracin | HMDB | | Ak-tracin | HMDB | | Altracin | HMDB | | Baci im | HMDB | | Baci-im | HMDB | | Bacitin | HMDB | | Bacitracin dioptic brand | HMDB | | Bacitracin lee brand | HMDB | | Bacitracin, zinc | HMDB | | Ciba vision brand OF bacitracin | HMDB | | Dioptic brand OF bacitracin | HMDB | | Pharma tek brand OF bacitracin | HMDB |
|
|---|
| Chemical Formula | C66H103N17O16S |
|---|
| Average Molecular Mass | 1422.693 g/mol |
|---|
| Monoisotopic Mass | 1421.749 g/mol |
|---|
| CAS Registry Number | 1405-87-4 |
|---|
| IUPAC Name | (4R)-4-[(2S)-2-{[(4R)-2-[(1S,2S)-1-amino-2-methylbutyl]-4,5-dihydro-1,3-thiazol-4-yl]formamido}-4-methylpentanamido]-4-{[(1S,2S)-1-{[(3S,6R,9S,12R,15S,18R,21S)-18-(3-aminopropyl)-12-benzyl-15-[(2S)-butan-2-yl]-3-(carbamoylmethyl)-6-(carboxymethyl)-9-(1H-imidazol-4-ylmethyl)-2,5,8,11,14,17,20-heptaoxo-1,4,7,10,13,16,19-heptaazacyclopentacosan-21-yl]carbamoyl}-2-methylbutyl]carbamoyl}butanoic acid |
|---|
| Traditional Name | baciguent |
|---|
| SMILES | CC[C@H](C)[C@H](N)C1=N[C@@H](CS1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H]1CCCCNC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](CC(O)=O)NC(=O)[C@H](CC2=CNC=N2)NC(=O)[C@@H](CC2=CC=CC=C2)NC(=O)[C@@H](NC(=O)[C@@H](CCCN)NC1=O)[C@@H](C)CC |
|---|
| InChI Identifier | InChI=1S/C66H103N17O16S/c1-9-35(6)52(69)66-81-48(32-100-66)63(97)76-43(26-34(4)5)59(93)74-42(22-23-50(85)86)58(92)83-53(36(7)10-2)64(98)75-40-20-15-16-25-71-55(89)46(29-49(68)84)78-62(96)47(30-51(87)88)79-61(95)45(28-39-31-70-33-72-39)77-60(94)44(27-38-18-13-12-14-19-38)80-65(99)54(37(8)11-3)82-57(91)41(21-17-24-67)73-56(40)90/h12-14,18-19,31,33-37,40-48,52-54H,9-11,15-17,20-30,32,67,69H2,1-8H3,(H2,68,84)(H,70,72)(H,71,89)(H,73,90)(H,74,93)(H,75,98)(H,76,97)(H,77,94)(H,78,96)(H,79,95)(H,80,99)(H,82,91)(H,83,92)(H,85,86)(H,87,88)/t35-,36-,37-,40-,41+,42+,43-,44+,45-,46-,47+,48-,52-,53-,54-/m0/s1 |
|---|
| InChI Key | CLKOFPXJLQSYAH-ABRJDSQDSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as polypeptides. These are peptides containing ten or more amino acid residues. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic Polymers |
|---|
| Class | Polypeptides |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Polypeptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Polypeptide
- Cyclic alpha peptide
- Glutamic acid or derivatives
- Leucine or derivatives
- Isoleucine or derivatives
- Macrolactam
- N-acyl-alpha amino acid or derivatives
- Alpha-amino acid amide
- N-substituted-alpha-amino acid
- Alpha-amino acid or derivatives
- Imidazolyl carboxylic acid derivative
- Thiazolecarboxamide
- Fatty amide
- N-acyl-amine
- Benzenoid
- Dicarboxylic acid or derivatives
- Monocyclic benzene moiety
- Fatty acyl
- Heteroaromatic compound
- Imidazole
- Azole
- Thiazole
- Meta-thiazoline
- Primary carboxylic acid amide
- Secondary carboxylic acid amide
- Carboxamide group
- Amino acid
- Lactam
- Amino acid or derivatives
- Azacycle
- Organoheterocyclic compound
- Carboxylic acid derivative
- Carboxylic acid
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Amine
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Organic nitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0f79-1228910011-797a0ade3d94224122c6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dr-4944200011-d12f20c9baa9394318f5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-9632100000-f2bc96584ade64ddae6f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0wb9-1228900000-f1be165ec00c831482ae | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0zgm-6249600021-e9dab8ea8d4bc2a73519 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05o3-9766400021-36bafa1e5409665b9d8a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0fk9-0003920000-17ed06ed1358f2553f33 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0nmi-0109400002-a4e62e7e2b6f20ebb356 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fsl-9743300001-ee63bf15f5f7e08b0d0d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0001900000-898dbfd2300b1dfad2bc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a7i-1019110000-9b9ef93049647047beb0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05mn-6966100000-81bb35604ad0986abff2 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB00626 |
|---|
| HMDB ID | HMDB0014764 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Bacitracin |
|---|
| Chemspider ID | 9084687 |
|---|
| ChEBI ID | 35862 |
|---|
| PubChem Compound ID | 10909430 |
|---|
| Kegg Compound ID | C15482 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|