| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:18:32 UTC |
|---|
| Update Date | 2016-11-09 01:17:25 UTC |
|---|
| Accession Number | CHEM022050 |
|---|
| Identification |
|---|
| Common Name | 5-Hydroxyfluvastatin |
|---|
| Class | Small Molecule |
|---|
| Description | 5-Hydroxyfluvastatin is only found in individuals that have used or taken Fluvastatin. 5-Hydroxyfluvastatin is a metabolite of Fluvastatin. 5-hydroxyfluvastatin belongs to the family of Indoles. These are compounds containing an indole moiety, which consists of pyrrole ring fused to benzene to form 2,3-benzopyrrole. |
|---|
| Contaminant Sources | - HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
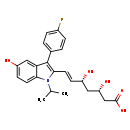
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (3S,5R,6E)-7-[3-(4-Fluorophenyl)-5-hydroxy-1-(propan-2-yl)-1H-indol-2-yl]-3,5-dihydroxyhept-6-enoate | HMDB |
|
|---|
| Chemical Formula | C24H26FNO5 |
|---|
| Average Molecular Mass | 427.465 g/mol |
|---|
| Monoisotopic Mass | 427.180 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (3S,5R,6E)-7-[3-(4-fluorophenyl)-5-hydroxy-1-(propan-2-yl)-1H-indol-2-yl]-3,5-dihydroxyhept-6-enoic acid |
|---|
| Traditional Name | (3S,5R,6E)-7-[3-(4-fluorophenyl)-5-hydroxy-1-isopropylindol-2-yl]-3,5-dihydroxyhept-6-enoic acid |
|---|
| SMILES | CC(C)N1C(\C=C\[C@H](O)C[C@H](O)CC(O)=O)=C(C2=CC=C(F)C=C2)C2=CC(O)=CC=C12 |
|---|
| InChI Identifier | InChI=1S/C24H26FNO5/c1-14(2)26-21-9-7-18(28)12-20(21)24(15-3-5-16(25)6-4-15)22(26)10-8-17(27)11-19(29)13-23(30)31/h3-10,12,14,17,19,27-29H,11,13H2,1-2H3,(H,30,31)/b10-8+/t17-,19-/m0/s1 |
|---|
| InChI Key | YCXJQQVRCVXLTP-CLFQVBOOSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylpyrroles. These are polycyclic aromatic compounds containing a benzene ring linked to a pyrrole ring through a CC or CN bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyrroles |
|---|
| Sub Class | Substituted pyrroles |
|---|
| Direct Parent | Phenylpyrroles |
|---|
| Alternative Parents | |
|---|
| Substituents | - 3-phenylpyrrole
- N-alkylindole
- Hydroxyindole
- Indole or derivatives
- Medium-chain hydroxy acid
- Indole
- Medium-chain fatty acid
- 1-hydroxy-2-unsubstituted benzenoid
- Hydroxy fatty acid
- Heterocyclic fatty acid
- Halogenated fatty acid
- Beta-hydroxy acid
- Halobenzene
- Fluorobenzene
- Unsaturated fatty acid
- Monocyclic benzene moiety
- Hydroxy acid
- Benzenoid
- Fatty acid
- Fatty acyl
- Aryl halide
- Aryl fluoride
- Heteroaromatic compound
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Azacycle
- Monocarboxylic acid or derivatives
- Organooxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Alcohol
- Carbonyl group
- Organic oxide
- Organopnictogen compound
- Organonitrogen compound
- Organofluoride
- Organohalogen compound
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-006x-8059300000-0d73c44045067200f971 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-004i-7210459000-0918ac631ebb1273edb4 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03dl-0008900000-c5e0e4bf3e8396ad0a8e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03xu-2019200000-0a6ba4e10d0db4844a91 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014u-8297000000-ed1264652f90059b214c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-1006900000-9be592465f9bec1dc2e4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0c09-6109200000-8d8dfab65a682728d55a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9025000000-c4c2ef8893a0493449c9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0004900000-635f4ca0b7765dfe5fce | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-022i-2009000000-e78cb6d9baef6646f252 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-5095000000-987d500522d5b7eb92cb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01t9-0003900000-b51e7721762fc8681a0b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01ox-0059500000-ab87194f6c7a0ae608df | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0uec-0092000000-ac6f4a91df0841abae3d | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0014039 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 30776730 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 124518231 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|