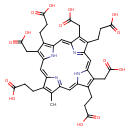
Identification Common Name Heptacarboxylporphyrin I Class Small Molecule Description Heptacarboxylporphyrin I is a bile product that arises from the conversion of Heptacarboxylporphyrinogen I to Heptacarboxylporphyrin I by porphyrinogen carboxy-lyase (EC 4.1.1.37). [HMDB] Contaminant Sources FooDB Chemicals
HMDB Contaminants - Urine Contaminant Type Not Available Chemical Structure Synonyms Value Source Heptacarboxylic acid porphyrin I HMDB Heptaporphyrin HMDB 3-[9,14,19-Tris(2-carboxyethyl)-10,15,20-tris(carboxymethyl)-5-methyl-21,22,23,24-tetraazapentacyclo[16.2.1.1³,⁶.1⁸,¹¹.1¹³,¹⁶]tetracosa-1,3(24),4,6,8,10,12,14,16(22),17,19-undecaen-4-yl]propanoate HMDB
Chemical Formula C39 H38 N4 O14 Average Molecular Mass 786.737 g/mol Monoisotopic Mass 786.238 g/mol CAS Registry Number 65406-45-3 IUPAC Name 3-[9,14,19-tris(2-carboxyethyl)-10,15,20-tris(carboxymethyl)-5-methyl-21,22,23,24-tetraazapentacyclo[16.2.1.1^{3,6}.1^{8,11}.1^{13,16}]tetracosa-1(20),2,4,6(24),7,9,11,13(22),14,16,18-undecaen-4-yl]propanoic acid Traditional Name 3-[9,14,19-tris(2-carboxyethyl)-10,15,20-tris(carboxymethyl)-5-methyl-21,22,23,24-tetraazapentacyclo[16.2.1.1^{3,6}.1^{8,11}.1^{13,16}]tetracosa-1(20),2,4,6(24),7,9,11,13(22),14,16,18-undecaen-4-yl]propanoic acid SMILES CC1=C(CCC(O)=O)\C2=C\C3=C(CC(O)=O)C(CCC(O)=O)=C(N3)\C=C3/N=C(/C=C4\N\C(=C/C1=N2)C(CCC(O)=O)=C4CC(O)=O)C(CCC(O)=O)=C3CC(O)=O InChI Identifier InChI=1S/C39H38N4O14/c1-17-18(2-6-33(44)45)26-14-30-23(11-38(54)55)20(4-8-35(48)49)28(42-30)16-32-24(12-39(56)57)21(5-9-36(50)51)29(43-32)15-31-22(10-37(52)53)19(3-7-34(46)47)27(41-31)13-25(17)40-26/h13-16,41-42H,2-12H2,1H3,(H,44,45)(H,46,47)(H,48,49)(H,50,51)(H,52,53)(H,54,55)(H,56,57)/b25-13-,26-14-,27-13-,28-16-,29-15-,30-14-,31-15-,32-16- InChI Key GWTVAIDNCPVMLP-YBWGHNILSA-N Spectra Spectra Spectrum Type Description Splash Key View Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_5) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_6) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_7) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_8) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_9) - 70eV, Positive Not Available Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-106r-0000000900-1a5659367722a3b91d07 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-0ab9-0000002900-85dfdd282818a628f93a Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-053r-0000009700-49b4b052511c4ae6e5ab Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-00xu-0000000900-ab85973f83598078c240 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-00di-0000001900-1a9755eb8354eff16f28 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-00di-2000000900-453451d4dc4aab7fd692 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-00xs-0000002900-7fb9688f9c1ee33b1308 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-00aj-0000009600-c6f4c536d4eb7fbe2584 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-003s-0000009500-95d5d3990319dc7e8707 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-00dj-0000006900-3a85eeb4b06df50b7894 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-006t-0000009700-fa3a78fcf7166dc4eeb0 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-00kb-0000009100-6f56ce92b761bddef4bd Spectrum