Identification Common Name Sulfolithocholylglycine Class Small Molecule Description Contaminant Sources FooDB Chemicals
HMDB Contaminants - Feces
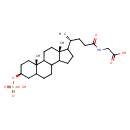
HMDB Contaminants - Urine Contaminant Type Not Available Chemical Structure Synonyms Value Source Sulpholithocholylglycine Generator 2-{[(4R)-4-[(2S,5S,15R)-2,15-dimethyl-5-(sulfooxy)tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-1-hydroxypentylidene]amino}acetate HMDB 2-{[(4R)-4-[(2S,5S,15R)-2,15-dimethyl-5-(sulphooxy)tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-1-hydroxypentylidene]amino}acetate HMDB 2-{[(4R)-4-[(2S,5S,15R)-2,15-dimethyl-5-(sulphooxy)tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]-1-hydroxypentylidene]amino}acetic acid HMDB GLCA-sul HMDB Glycine amidated HMDB Glycolithocholate 3-sulfate HMDB Glycolithocholate 3-sulphate HMDB Glycolithocholate sulfate HMDB Glycolithocholate sulphate HMDB Glycolithocholic acid 3-sulfate HMDB Glycolithocholic acid 3-sulphate HMDB Glycolithocholic acid sulfate HMDB Glycolithocholic acid sulphate HMDB LCA-sul HMDB Sulfoglycolithocholate HMDB Sulfoglycolithocholic acid HMDB
Chemical Formula C26 H43 NO7 S Average Molecular Mass 513.687 g/mol Monoisotopic Mass 513.276 g/mol CAS Registry Number 15324-64-8 IUPAC Name 2-[(4R)-4-[(2S,5S,15R)-2,15-dimethyl-5-(sulfooxy)tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanamido]acetic acid Traditional Name [(4R)-4-[(2S,5S,15R)-2,15-dimethyl-5-(sulfooxy)tetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadecan-14-yl]pentanamido]acetic acid SMILES C[C@H](CCC(=O)NCC(O)=O)C1CCC2C3CCC4C[C@H](CC[C@]4(C)C3CC[C@]12C)OS(O)(=O)=O InChI Identifier InChI=1S/C26H43NO7S/c1-16(4-9-23(28)27-15-24(29)30)20-7-8-21-19-6-5-17-14-18(34-35(31,32)33)10-12-25(17,2)22(19)11-13-26(20,21)3/h16-22H,4-15H2,1-3H3,(H,27,28)(H,29,30)(H,31,32,33)/t16-,17?,18+,19?,20?,21?,22?,25+,26-/m1/s1 InChI Key FHXBAFXQVZOILS-JWOIRZCRSA-N