| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:09:38 UTC |
|---|
| Update Date | 2016-11-09 01:17:20 UTC |
|---|
| Accession Number | CHEM021671 |
|---|
| Identification |
|---|
| Common Name | Biotin sulfone |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - FooDB Chemicals
- HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
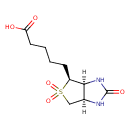
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Bisnorbiotin methyl ketone | HMDB | | [3AS-(3aa,4b,6aa)]-tetrahydro-4-(3-oxobutyl)-1H-thieno[3,4-D]imidazol-2(3H)-one | HMDB | | 5,5-Dioxide biotin | ChEBI | | [3AS-(3aa,4b,6aa)]-5,5-dioxide-hexahydro-2-oxo-1H-thieno[3,4-D]imidazole-4-pentanoic acid | ChEBI | | Biotin sulphone | Generator | | [3AS-(3aa,4b,6aa)]-5,5-dioxide-hexahydro-2-oxo-1H-thieno[3,4-D]imidazole-4-pentanoate | Generator | | [3AS-(3aa,4b,6aa)]- 5,5-dioxide-hexahydro-2-oxo-1H-thieno[3,4-D]imidazole-4-pentanoate | HMDB | | [3AS-(3aa,4b,6aa)]- 5,5-dioxide-hexahydro-2-oxo-1H-thieno[3,4-D]imidazole-4-pentanoic acid | HMDB | | 5-((3AS,4S,6ar)-5,5-dioxido-2-oxohexahydro-1H-thieno(3,4-D)imidazol-4-yl)pentanoic acid | MeSH |
|
|---|
| Chemical Formula | C10H16N2O5S |
|---|
| Average Molecular Mass | 276.309 g/mol |
|---|
| Monoisotopic Mass | 276.078 g/mol |
|---|
| CAS Registry Number | 40720-05-6 |
|---|
| IUPAC Name | 5-[(3aS,4S,6aR)-2,5,5-trioxo-hexahydro-1H-5λ⁶-thieno[3,4-d]imidazolidin-4-yl]pentanoic acid |
|---|
| Traditional Name | 5-[(3aS,4S,6aR)-2,5,5-trioxo-hexahydro-5λ⁶-thieno[3,4-d]imidazolidin-4-yl]pentanoic acid |
|---|
| SMILES | [H][C@]12CS(=O)(=O)[C@@H](CCCCC(O)=O)[C@@]1([H])NC(=O)N2 |
|---|
| InChI Identifier | InChI=1S/C10H16N2O5S/c13-8(14)4-2-1-3-7-9-6(5-18(7,16)17)11-10(15)12-9/h6-7,9H,1-5H2,(H,13,14)(H2,11,12,15)/t6-,7-,9-/m0/s1 |
|---|
| InChI Key | QPFQYMONYBAUCY-ZKWXMUAHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as thienoimidazolidines. These are heterocyclic compounds containing a thiophene ring fused to an imidazolidine ring. Thiophene is 5-membered ring consisting of four carbon atoms and one sulfur atom. Imidazolidine is 5-membered saturated ring of three carbon atoms, and two nitrogen centers at the 1- and 3-positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Thienoimidazolidines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Thienoimidazolidines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Thienoimidazolidine
- Imidazolidinone
- Imidazolidine
- Thiolane
- Thiophene
- Ketone
- Carbonic acid derivative
- Urea
- Thioether
- Dialkylthioether
- Azacycle
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Organic oxygen compound
- Organic nitrogen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-9440000000-1c52a504c730b15049b3 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-00xr-6970000000-30089da78cf0ca4f5efa | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-056r-0090000000-c44a2d9a678137742bbd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0532-2790000000-73413860cfdb8bf97ca7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0043-9620000000-d431dda06f942c88bb29 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-2390000000-adaa3c0f7ad790361a80 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052g-9340000000-f26b2be172eef38fc87b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9100000000-ee2350bbdc53d81139e0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0090000000-c08757af0593294271ee | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a6r-0190000000-6e408e98cc5ac169b089 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-9300000000-5d1f8e0280a38b7bf585 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0090000000-014d3cba016fc21f50ed | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-056r-0090000000-2d6335dc30bf7a96f43c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0059-5490000000-d899e19c8e3d05d6dcc1 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0004822 |
|---|
| FooDB ID | FDB023430 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | 7084 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 13628421 |
|---|
| ChEBI ID | 89480 |
|---|
| PubChem Compound ID | 21252323 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|