| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 18:08:53 UTC |
|---|
| Update Date | 2016-11-09 01:17:20 UTC |
|---|
| Accession Number | CHEM021638 |
|---|
| Identification |
|---|
| Common Name | N1-Acetylspermidine |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | - FooDB Chemicals
- HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
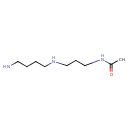
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-N-Acetylspermidine | ChEBI | | An N-acetylspermidine | HMDB | | N(1)-Acetylspermidine | HMDB | | N-(3-((4-Aminobutyl)amino)propyl)-acetamide | HMDB | | N-(3-((4-Aminobutyl)amino)propyl)acetamide | HMDB | | N-Acetylspermidine | HMDB | | N1-Acetylspermidine | ChEBI |
|
|---|
| Chemical Formula | C9H21N3O |
|---|
| Average Molecular Mass | 187.283 g/mol |
|---|
| Monoisotopic Mass | 187.168 g/mol |
|---|
| CAS Registry Number | 14278-49-0 |
|---|
| IUPAC Name | N-{3-[(4-aminobutyl)amino]propyl}acetamide |
|---|
| Traditional Name | N1-acetylspermidine |
|---|
| SMILES | CC(=O)NCCCNCCCCN |
|---|
| InChI Identifier | InChI=1S/C9H21N3O/c1-9(13)12-8-4-7-11-6-3-2-5-10/h11H,2-8,10H2,1H3,(H,12,13) |
|---|
| InChI Key | MQTAVJHICJWXBR-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as carboximidic acids. These are organic acids with the general formula RC(=N)-OH (R=H, organic group). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboximidic acids and derivatives |
|---|
| Sub Class | Carboximidic acids |
|---|
| Direct Parent | Carboximidic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Secondary amine
- Secondary aliphatic amine
- Carboximidic acid
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Amine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00ec-9300000000-435442e25431afc9ca94 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00g1-1900000000-25a42a25b21ff785892b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05dj-6900000000-12de75772a08ca08852a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05fu-9200000000-2dfa5d648a84baff3e54 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-1900000000-90283b76b9478101a593 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052o-5900000000-4cac7a34f5b572449747 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9000000000-46e81717ccd7a43b7c55 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0079-3900000000-7dcca0a83898d18d48ff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ab9-9300000000-db42164e7ae02cc3565e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05gi-9000000000-b73b9a6524aaa490eb70 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0900000000-b573184691a2508c0c96 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4r-9700000000-683029d89d5350729ba4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9000000000-236e795417fce314ee2b | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0001276 |
|---|
| FooDB ID | FDB022530 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | CPD-568 |
|---|
| METLIN ID | 3323 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 482 |
|---|
| ChEBI ID | 17927 |
|---|
| PubChem Compound ID | 496 |
|---|
| Kegg Compound ID | C00612 |
|---|
| YMDB ID | YMDB00366 |
|---|
| ECMDB ID | ECMDB01276 |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Lurdes M; Almeida S; Grehn L; Ragnarsson U Facile synthesis of monoacetylated spermidines, illustrating selective deacetylation and application of a common precursor. Acta chemica Scandinavica (Copenhagen, Denmark : 1989) (1989), 43(10), 990-4. | | 2. Hrushesky WJ, Merdink J, Abdel-Monem MM: Circadian rhythmicity of polyamine urinary excretion. Cancer Res. 1983 Aug;43(8):3944-7. | | 3. Seiler N, Graham A, Bartholeyns J: Enhanced urinary excretion of N1-acetylspermidine and the presence of tumors. Cancer Res. 1981 Apr;41(4):1572-3. | | 4. Abdel-Monem MM, Merdink JL, Theologides A: Urinary excretion of monoacetyl polyamines in patients with non-Hodgkin's lymphoma. Cancer Res. 1982 May;42(5):2097-8. | | 5. Smith RG, Bartos D, Bartos F, Grettie DP, Frick W, Campbell RA, Daves GD Jr: 1-N-Acetylspermidine: occurrence in normal human serum. Biomed Mass Spectrom. 1978 Sep;5(9):515-7. | | 6. Ruggeri P, Lagana G, Bellocco E, Fabiano C, Leonaldi R, Forino D: Polyamines biosynthesis and oxidation in free-living amoebae. Amino Acids. 2004 Dec;27(3-4):367-72. Epub 2004 Oct 15. | | 7. https://www.ncbi.nlm.nih.gov/pubmed/?term=22770225 | | 8. https://www.ncbi.nlm.nih.gov/pubmed/?term=8446047 |
|
|---|