| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 06:53:01 UTC |
|---|
| Update Date | 2016-11-09 01:16:09 UTC |
|---|
| Accession Number | CHEM020177 |
|---|
| Identification |
|---|
| Common Name | Flavopiridol hydrochloride |
|---|
| Class | Small Molecule |
|---|
| Description | A hydrochloride salt resulting from the formal reaction of equimolar amounts of alvocidib and hydrogen chloride. A cyclin-dependent kinase 9 (CDK9) inhibitor, it has been studied for the treatment of acute myeloid leukaemia, arthritis and atherosclerotic plaque formation. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
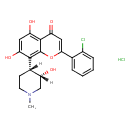
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (3S,4R)-4-[2-(2-Chlorophenyl)-5,7-dihydroxy-4-oxo-4H-chromen-8-yl]-3-hydroxy-1-methylpiperidinium chloride | ChEBI | | Alvocidib HCL | ChEBI | | HL 275 | ChEBI | | HL-275 | ChEBI | | HMR 1275 | ChEBI | | HMR-1275 | ChEBI | | L 86 8275 | ChEBI | | L86-8275 | ChEBI | | Alvocidib | MeSH | | Flavopiridol | MeSH | | (-)cis-5,7-Dihydroxy-2-(2-chlorophenyl)-8-(4-(3-hydroxy-1-methyl)piperidinyl)-4H-1-benzopyran-4-one | MeSH |
|
|---|
| Chemical Formula | C21H21Cl2NO5 |
|---|
| Average Molecular Mass | 438.301 g/mol |
|---|
| Monoisotopic Mass | 437.080 g/mol |
|---|
| CAS Registry Number | 131740-09-5 |
|---|
| IUPAC Name | 2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S,4R)-3-hydroxy-1-methylpiperidin-4-yl]-4H-chromen-4-one hydrochloride |
|---|
| Traditional Name | flavopiridol hydrochloride |
|---|
| SMILES | Cl.[H][C@@]1(O)CN(C)CC[C@]1([H])C1=C2OC(=CC(=O)C2=C(O)C=C1O)C1=CC=CC=C1Cl |
|---|
| InChI Identifier | InChI=1S/C21H20ClNO5.ClH/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22;/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3;1H/t12-,17+;/m0./s1 |
|---|
| InChI Key | LGMSNQNWOCSPIK-LWHGMNCYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as flavones. These are flavonoids with a structure based on the backbone of 2-phenylchromen-4-one (2-phenyl-1-benzopyran-4-one). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Flavones |
|---|
| Direct Parent | Flavones |
|---|
| Alternative Parents | |
|---|
| Substituents | - 5-hydroxyflavonoid
- 7-hydroxyflavonoid
- Flavone
- Hydroxyflavonoid
- Phenylpiperidine
- Chromone
- Benzopyran
- 1-benzopyran
- 1-hydroxy-2-unsubstituted benzenoid
- Chlorobenzene
- Halobenzene
- Pyranone
- Aralkylamine
- Benzenoid
- Pyran
- Piperidine
- Aryl chloride
- Aryl halide
- Monocyclic benzene moiety
- Heteroaromatic compound
- Vinylogous acid
- Tertiary aliphatic amine
- Tertiary amine
- Secondary alcohol
- 1,2-aminoalcohol
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Alcohol
- Organic oxide
- Organopnictogen compound
- Organic nitrogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Organohalogen compound
- Organochloride
- Organonitrogen compound
- Organooxygen compound
- Amine
- Hydrochloride
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0000900000-827dd2143560ad1a3d84 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-0000900000-827dd2143560ad1a3d84 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-0000900000-827dd2143560ad1a3d84 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0000900000-4daedc3c2bba43d6834a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0000900000-4daedc3c2bba43d6834a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000i-0000900000-4daedc3c2bba43d6834a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DBSALT000800 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 90998 |
|---|
| PubChem Compound ID | 9910986 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|