| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 06:52:52 UTC |
|---|
| Update Date | 2016-11-09 01:16:09 UTC |
|---|
| Accession Number | CHEM020173 |
|---|
| Identification |
|---|
| Common Name | Doramapimod |
|---|
| Class | Small Molecule |
|---|
| Description | Doramapimod is a P38 MAP kinase inhibitor. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
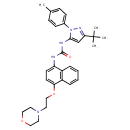
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-(3-(1,1-Dimethylethyl)-1-(4-methylphenyl)-1H-pyrazol-5-yl)-3-(4-(2-(morpholin-4-yl)ethoxy)naphthalen-1-yl)urea | ChEBI | | 1-[3-Tert-butyl-1-(4-methylphenyl)-1H-pyrazol-5-yl]-3-[4-(2-morpholin-4-ylethoxy)naphthalen-1-yl]urea | ChEBI | | BIRB 796 | ChEBI | | BIRB-796 | ChEBI | | 1-(5-Tert-butyl-2-P-tolyl-2H-pyrazol-3-yl)-3(4-(2-morpholin-4-yl-ethoxy)naph- thalen-1-yl)urea | MeSH | | BIRB796 | MeSH |
|
|---|
| Chemical Formula | C31H37N5O3 |
|---|
| Average Molecular Mass | 527.657 g/mol |
|---|
| Monoisotopic Mass | 527.290 g/mol |
|---|
| CAS Registry Number | 285983-48-4 |
|---|
| IUPAC Name | 3-[3-tert-butyl-1-(4-methylphenyl)-1H-pyrazol-5-yl]-1-{4-[2-(morpholin-4-yl)ethoxy]naphthalen-1-yl}urea |
|---|
| Traditional Name | doramapimod |
|---|
| SMILES | CC1=CC=C(C=C1)N1N=C(C=C1NC(=O)NC1=CC=C(OCCN2CCOCC2)C2=C1C=CC=C2)C(C)(C)C |
|---|
| InChI Identifier | InChI=1S/C31H37N5O3/c1-22-9-11-23(12-10-22)36-29(21-28(34-36)31(2,3)4)33-30(37)32-26-13-14-27(25-8-6-5-7-24(25)26)39-20-17-35-15-18-38-19-16-35/h5-14,21H,15-20H2,1-4H3,(H2,32,33,37) |
|---|
| InChI Key | MVCOAUNKQVWQHZ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylpyrazoles. Phenylpyrazoles are compounds containing a phenylpyrazole skeleton, which consists of a pyrazole bound to a phenyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Azoles |
|---|
| Sub Class | Pyrazoles |
|---|
| Direct Parent | Phenylpyrazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenylpyrazole
- Naphthalene
- Alkyl aryl ether
- Toluene
- Monocyclic benzene moiety
- Morpholine
- Oxazinane
- Benzenoid
- Heteroaromatic compound
- Carbonic acid derivative
- Urea
- Tertiary aliphatic amine
- Tertiary amine
- Dialkyl ether
- Ether
- Oxacycle
- Azacycle
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Amine
- Organic nitrogen compound
- Carbonyl group
- Organonitrogen compound
- Organooxygen compound
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-003r-0290030000-d0c70178952d29bca8f1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0390000000-de29559d9fb21dd89151 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001r-1920000000-aa8511a5614cb8d8b3bd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0190130000-ed59feaab932c7942064 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-05di-1692310000-88a286b95902fd4c9ada | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0570-3960000000-5fcad3e9376063d2d631 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0000190000-ea512a35acec75d3c82f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0350390000-387b06235c3737d20c49 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03k9-9630300000-fb008a1915b63a1f6a14 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0090030000-a6c0b381b2ad36be4b34 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-1394860000-238ab4475ee8964f4ed8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01t9-1691210000-ff2da628455146322bc4 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB03044 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 40953 |
|---|
| PubChem Compound ID | 156422 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|