| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 06:51:37 UTC |
|---|
| Update Date | 2016-11-09 01:16:08 UTC |
|---|
| Accession Number | CHEM020161 |
|---|
| Identification |
|---|
| Common Name | Selumetinib |
|---|
| Class | Small Molecule |
|---|
| Description | A member of the class of benzimidazoles that is 1-methyl-1H-benzimidazole which is substituted at positions 4, 5, and 6 by fluorine, (4-bromo-2-chlorophenyl)amino, and N-(2-hydroxyethoxy)aminocarbonyl groups, respectively. It is a MEK1 and MEK2 inhibitor. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
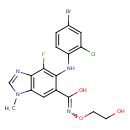
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| ARRY 142886 | ChEBI | | ARRY-142886 | ChEBI | | ARRY142886 | ChEBI | | AZD 6244 | ChEBI | | AZD-6244 | ChEBI | | AZD6244 | ChEBI | | Selumetinibum | ChEBI | | Selumetinib | MeSH |
|
|---|
| Chemical Formula | C17H15BrClFN4O3 |
|---|
| Average Molecular Mass | 457.680 g/mol |
|---|
| Monoisotopic Mass | 456.000 g/mol |
|---|
| CAS Registry Number | 606143-52-6 |
|---|
| IUPAC Name | 5-[(4-bromo-2-chlorophenyl)amino]-4-fluoro-N-(2-hydroxyethoxy)-1-methyl-1H-1,3-benzodiazole-6-carboximidic acid |
|---|
| Traditional Name | 6-[(4-bromo-2-chlorophenyl)amino]-7-fluoro-N-(2-hydroxyethoxy)-3-methyl-1,3-benzodiazole-5-carboximidic acid |
|---|
| SMILES | CN1C=NC2=C1C=C(C(O)=NOCCO)C(NC1=C(Cl)C=C(Br)C=C1)=C2F |
|---|
| InChI Identifier | InChI=1S/C17H15BrClFN4O3/c1-24-8-21-16-13(24)7-10(17(26)23-27-5-4-25)15(14(16)20)22-12-3-2-9(18)6-11(12)19/h2-3,6-8,22,25H,4-5H2,1H3,(H,23,26) |
|---|
| InChI Key | CYOHGALHFOKKQC-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzimidazoles. These are organic compounds containing a benzene ring fused to an imidazole ring (five member ring containing a nitrogen atom, 4 carbon atoms, and two double bonds). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzimidazoles |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzimidazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzimidazole
- Aniline or substituted anilines
- Bromobenzene
- Chlorobenzene
- Halobenzene
- Aryl bromide
- Aryl chloride
- Aryl fluoride
- Aryl halide
- Monocyclic benzene moiety
- Benzenoid
- N-substituted imidazole
- Azole
- Heteroaromatic compound
- Vinylogous amide
- Imidazole
- Amino acid or derivatives
- Azacycle
- Carboxylic acid derivative
- Secondary amine
- Organooxygen compound
- Primary alcohol
- Organohalogen compound
- Organobromide
- Amine
- Hydrocarbon derivative
- Alcohol
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Organochloride
- Organofluoride
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a6r-5003900000-ad7092eb9e18e8c63f94 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-2009100000-3858abbf1afdf7e8f6f3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-002b-9006000000-a56224b65db84bc5eb20 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-1002900000-035bcb0c187f51cde944 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udl-4009400000-abdf560a6933bf91112d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ug0-4229000000-d09a5ee27c45ba878051 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0000900000-73d7e6711f3af0f387fc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-053r-0009800000-9075ace0cdf9bf6369a0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-0139100000-acd888a1944907e50ac2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0001900000-37aa71a8f8f030f74806 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f6x-4009500000-567c825d54ceb9e7e6a4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-3009000000-6f51161796301c9b94b6 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB11689 |
|---|
| HMDB ID | HMDB0304858 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Selumetinib |
|---|
| Chemspider ID | 8303141 |
|---|
| ChEBI ID | 90227 |
|---|
| PubChem Compound ID | 10127622 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|