| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 06:41:53 UTC |
|---|
| Update Date | 2016-11-09 01:16:07 UTC |
|---|
| Accession Number | CHEM020011 |
|---|
| Identification |
|---|
| Common Name | Troxacitabine |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Troxacitabine, 2R(-)-cis-isomer | MeSH | | Troxacitabine, 2R(-)-trans-isomer | MeSH | | Troxacitabine, cis-(+-)-isomer | MeSH | | HMD-Cytosine | MeSH | | beta-L-Dioxolane-cytidine | MeSH | | 2R(-)-trans-HMD-Cytosine | MeSH | | Troxatyl | MeSH | | Troxacitabine, 2S(-)-trans-isomer | MeSH | | 1-(2-(Hydroxymethyl)-1,3-dioxolan-4-yl)cytosine | MeSH | | 2R(-)-cis-HMD-Cytosine | MeSH | | Troxacitabine, trans-(+-)-isomer | MeSH |
|
|---|
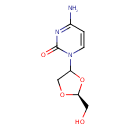
| Chemical Formula | C8H11N3O4 |
|---|
| Average Molecular Mass | 213.191 g/mol |
|---|
| Monoisotopic Mass | 213.075 g/mol |
|---|
| CAS Registry Number | 145918-75-8 |
|---|
| IUPAC Name | 4-amino-1-[(2S)-2-(hydroxymethyl)-1,3-dioxolan-4-yl]-1,2-dihydropyrimidin-2-one |
|---|
| Traditional Name | troxacitabine |
|---|
| SMILES | NC1=NC(=O)N(C=C1)C1CO[C@H](CO)O1 |
|---|
| InChI Identifier | InChI=1S/C8H11N3O4/c9-5-1-2-11(8(13)10-5)6-4-14-7(3-12)15-6/h1-2,6-7,12H,3-4H2,(H2,9,10,13)/t6?,7-/m0/s1 |
|---|
| InChI Key | RXRGZNYSEHTMHC-MLWJPKLSSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as nucleoside and nucleotide analogues. These are analogues of nucleosides and nucleotides. These include phosphonated nucleosides, C-glycosylated nucleoside bases, analogues where the sugar unit is a pyranose, and carbocyclic nucleosides, among others. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | Nucleoside and nucleotide analogues |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Nucleoside and nucleotide analogues |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aminopyrimidine
- Pyrimidone
- Hydropyrimidine
- Pyrimidine
- Imidolactam
- Meta-dioxolane
- Heteroaromatic compound
- Acetal
- Oxacycle
- Organoheterocyclic compound
- Azacycle
- Organic oxide
- Primary amine
- Primary alcohol
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Alcohol
- Amine
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0920000000-33e2c218b6cd8c8ecb44 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-4900000000-ad3ef4f6e28365a76fd0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03xs-9400000000-9da209dedebc5a80a131 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-1930000000-4514c625058f76b81704 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-02t9-3900000000-cd18ca920a28abd2118d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00kf-9100000000-4bba923b4fc236b7efae | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 151173 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|