| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 06:32:46 UTC |
|---|
| Update Date | 2016-11-09 01:16:04 UTC |
|---|
| Accession Number | CHEM019821 |
|---|
| Identification |
|---|
| Common Name | Pralatrexate |
|---|
| Class | Small Molecule |
|---|
| Description | A pteridine that is the N-4-benzoyl derivative of L-glutamic acid. Used for treatment of Peripheral T-Cell Lymphoma, an aggressive form of non-Hodgkins lymphoma. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
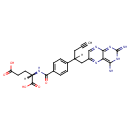
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2S)-2-((4-((1Rs)-1-((2,4-Diaminopteridin-6-yl)methyl)but-3-ynyl)benzoyl)amino)pentanedioic acid | ChEBI | | (2S)-2-({4-[1-(2,4-diaminopteridin-6-yl)pent-4-yn-2-yl]benzoyl}amino)pentanedioic acid | ChEBI | | 10-Propargyl-10-deazaaminopterin | ChEBI | | Folotyn | ChEBI | | HSDB 7786 | ChEBI | | N-(4-(1-((2,4-Diamino-6-pteridinyl)methyl)-3-butynyl)benzoyl)-L-glutamic acid | ChEBI | | Pralatrexato | ChEBI | | Pralatrexatum | ChEBI | | (2S)-2-((4-((1Rs)-1-((2,4-Diaminopteridin-6-yl)methyl)but-3-ynyl)benzoyl)amino)pentanedioate | Generator | | (2S)-2-({4-[1-(2,4-diaminopteridin-6-yl)pent-4-yn-2-yl]benzoyl}amino)pentanedioate | Generator | | N-(4-(1-((2,4-Diamino-6-pteridinyl)methyl)-3-butynyl)benzoyl)-L-glutamate | Generator | | Pralatrexic acid | Generator |
|
|---|
| Chemical Formula | C23H23N7O5 |
|---|
| Average Molecular Mass | 477.473 g/mol |
|---|
| Monoisotopic Mass | 477.176 g/mol |
|---|
| CAS Registry Number | 146464-95-1 |
|---|
| IUPAC Name | (2S)-2-({4-[1-(2,4-diaminopteridin-6-yl)pent-4-yn-2-yl]phenyl}formamido)pentanedioic acid |
|---|
| Traditional Name | pralatrexate |
|---|
| SMILES | NC1=NC2=NC=C(CC(CC#C)C3=CC=C(C=C3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)N=C2C(N)=N1 |
|---|
| InChI Identifier | InChI=1S/C23H23N7O5/c1-2-3-14(10-15-11-26-20-18(27-15)19(24)29-23(25)30-20)12-4-6-13(7-5-12)21(33)28-16(22(34)35)8-9-17(31)32/h1,4-7,11,14,16H,3,8-10H2,(H,28,33)(H,31,32)(H,34,35)(H4,24,25,26,29,30)/t14?,16-/m0/s1 |
|---|
| InChI Key | OGSBUKJUDHAQEA-WMCAAGNKSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzothiadiazoles. These are heterocyclic aromatic compounds containing a benzene ring fused to a thiadiazole ring. Thiadiazole is a five-membered aromatic heterocycle made up of one sulfur atom and two nitrogen atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzothiadiazoles |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzothiadiazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1,2,3-benzothiadiazole
- Thiobenzoic acid or derivatives
- Benzenoid
- Azole
- Thiadiazole
- Heteroaromatic compound
- Carbothioic s-ester
- Thiocarboxylic acid ester
- Azacycle
- Sulfenyl compound
- Thiocarboxylic acid or derivatives
- Carboxylic acid derivative
- Organopnictogen compound
- Organic oxygen compound
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-004i-1513900000-5596beb52832aa3566cc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01si-0002900000-6fc75eff8254786e9df5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0106900000-5ac9be915593c90e1ce4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0059-2698000000-a5acf2f9d1da2432962a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0000900000-b353523bc14906b8a722 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-003r-1203900000-e69e51466057378426a7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9425000000-129bfd939baa92b07749 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB06813 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Pralatrexate |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 71223 |
|---|
| PubChem Compound ID | 148121 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|