| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 06:07:00 UTC |
|---|
| Update Date | 2016-11-09 01:15:59 UTC |
|---|
| Accession Number | CHEM019432 |
|---|
| Identification |
|---|
| Common Name | Fandosentan potassium salt |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
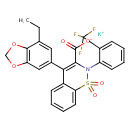
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Potassium 4-(7-ethyl-2H-1,3-benzodioxol-5-yl)-1,1-dioxo-2-[2-(trifluoromethyl)phenyl]-2H-1,2-benzothiazine-3-carboxylic acid | Generator | | PD-180988pd-180988-0016CI-1034fandosentan potassium | ChEMBL | | Potassium;4-(7-ethyl-1,3-benzodioxol-5-yl)-1,1-dioxo-2-[2-(trifluoromethyl)phenyl]-1$l^{6},2-benzothiazine-3-carboxylic acid | Generator | | Potassium 4-(7-ethyl-2H-1,3-benzodioxol-5-yl)-1,1-dioxo-2-[2-(trifluoromethyl)phenyl]-2H-1λ⁶,2-benzothiazine-3-carboxylic acid | Generator |
|
|---|
| Chemical Formula | C25H17F3KNO6S |
|---|
| Average Molecular Mass | 555.570 g/mol |
|---|
| Monoisotopic Mass | 555.037 g/mol |
|---|
| CAS Registry Number | 221246-12-4 |
|---|
| IUPAC Name | potassium 4-(7-ethyl-2H-1,3-benzodioxol-5-yl)-1,1-dioxo-2-[2-(trifluoromethyl)phenyl]-2H-1λ⁶,2-benzothiazine-3-carboxylate |
|---|
| Traditional Name | potassium 4-(7-ethyl-2H-1,3-benzodioxol-5-yl)-1,1-dioxo-2-[2-(trifluoromethyl)phenyl]-1λ⁶,2-benzothiazine-3-carboxylate |
|---|
| SMILES | [K+].CCC1=CC(=CC2=C1OCO2)C1=C(N(C2=CC=CC=C2C(F)(F)F)S(=O)(=O)C2=CC=CC=C12)C([O-])=O |
|---|
| InChI Identifier | InChI=1S/C25H18F3NO6S.K/c1-2-14-11-15(12-19-23(14)35-13-34-19)21-16-7-3-6-10-20(16)36(32,33)29(22(21)24(30)31)18-9-5-4-8-17(18)25(26,27)28;/h3-12H,2,13H2,1H3,(H,30,31);/q;+1/p-1 |
|---|
| InChI Key | MZWCHWWQSPPZRH-UHFFFAOYSA-M |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzothiazines. These are organic compounds containing a benzene fused to a thiazine ring (a six-membered ring with four carbon atoms, one nitrogen atom and one sulfur atom). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzothiazines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzothiazines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-amino acid or derivatives
- Trifluoromethylbenzene
- Benzothiazine
- Sulfanilide
- Benzodioxole
- Ortho-thiazine
- Monocyclic benzene moiety
- Organosulfonic acid amide
- Benzenoid
- Organic sulfonic acid or derivatives
- Organosulfonic acid or derivatives
- Carboxylic acid salt
- Acetal
- Organic metal halide
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Oxacycle
- Azacycle
- Organic alkali metal salt
- Organohalogen compound
- Organic oxygen compound
- Alkyl halide
- Hydrocarbon derivative
- Alkyl fluoride
- Carbonyl group
- Organic oxide
- Organofluoride
- Organonitrogen compound
- Organooxygen compound
- Organopnictogen compound
- Organic nitrogen compound
- Organic potassium salt
- Organic salt
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4r-0010190000-204dea667d6da5107863 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0294100000-83bd6771cab68c5d7311 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a5l-2970400000-0475b2d3d7b6c102adb5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udj-0000960000-56318371e691b4635e66 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0uka-0001940000-be3a0e3fda46f389ffc2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01vn-1590100000-54eb92995123bf6707b5 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 157573 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|