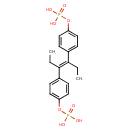| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 06:02:19 UTC |
|---|
| Update Date | 2016-11-09 01:15:58 UTC |
|---|
| Accession Number | CHEM019343 |
|---|
| Identification |
|---|
| Common Name | Diethylstilbestrol diphosphate |
|---|
| Class | Small Molecule |
|---|
| Description | Fosfestrol, sold under the brand name Honvan and also known as diethylstilbestrol diphosphate (DESDP), is an estrogen medication which is used in the treatment of prostate cancer in men. It is given by slow intravenous infusion once per day to once per week or by mouth once per day.Side effects of fosfestrol include nausea and vomiting, cardiovascular complications, blood clots, edema, and genital skin reactions, among others. Fosfestrol is an estrogen, and hence is an agonist of the estrogen receptor, the biological target of estrogens like estradiol. It acts as a prodrug of diethylstilbestrol.Fosfestrol was patented in 1941 and was introduced for medical use in 1955. It was previously marketed widely throughout the world, but now remains available in only a few countries. |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Diethylstilbestrol bisphosphate | Kegg | | Stilphostrol | Kegg | | Diethylstilbestrol bisphosphoric acid | Generator | | Fosfestrol, tetrasodium salt, (e)-isomer | MeSH | | Diethyldioxystilbene diphosphate | MeSH | | Diethylstilbestrol diphosphate | MeSH | | Difostilben | MeSH | | Fosfestrol, sodium salt, (e)-isomer | MeSH | | Kyorin | MeSH | | Fosfestrol, 99TC-labeled CPD | MeSH | | Fosfesterol | MeSH | | Fosfestrol, disodium salt, (e)-isomer | MeSH | | Honvan | MeSH | | ST-52 | MeSH | | Fostrolin | MeSH | | Stilbestrol diphosphate | MeSH | | Fosfestrol, (e)-isomer | MeSH | | Fostestrolum | MeSH | | Fosfestrol | KEGG | | Diethylstilbestrol diphosphoric acid | Generator | | [4-[(e)-4-(4-Phosphonooxyphenyl)hex-3-en-3-yl]phenyl] dihydrogen phosphoric acid | Generator | | {4-[(3E)-4-[4-(phosphonooxy)phenyl]hex-3-en-3-yl]phenoxy}phosphonate | Generator |
|
|---|
| Chemical Formula | C18H22O8P2 |
|---|
| Average Molecular Mass | 428.314 g/mol |
|---|
| Monoisotopic Mass | 428.079 g/mol |
|---|
| CAS Registry Number | 522-40-7 |
|---|
| IUPAC Name | {4-[(3E)-4-[4-(phosphonooxy)phenyl]hex-3-en-3-yl]phenoxy}phosphonic acid |
|---|
| Traditional Name | fosfestrol |
|---|
| SMILES | CC\C(=C(\CC)C1=CC=C(OP(O)(O)=O)C=C1)C1=CC=C(OP(O)(O)=O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C18H22O8P2/c1-3-17(13-5-9-15(10-6-13)25-27(19,20)21)18(4-2)14-7-11-16(12-8-14)26-28(22,23)24/h5-12H,3-4H2,1-2H3,(H2,19,20,21)(H2,22,23,24)/b18-17+ |
|---|
| InChI Key | NLORYLAYLIXTID-ISLYRVAYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as stilbenes. These are organic compounds containing a 1,2-diphenylethylene moiety. Stilbenes (C6-C2-C6 ) are derived from the common phenylpropene (C6-C3) skeleton building block. The introduction of one or more hydroxyl groups to a phenyl ring lead to stilbenoids. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Stilbenes |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Stilbenes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Stilbene
- Phenyl phosphate
- Aryl phosphate
- Aryl phosphomonoester
- Phenylpropane
- Phenoxy compound
- Benzenoid
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Monocyclic benzene moiety
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-3022900000-f0dd3283b1f6ee0ddc93 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-003r-2159500000-331fdc9897892806bfd0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001j-2193000000-9208d58eb1e03a31ada6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-9000300000-5cf1f4af7dbf70bfe431 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-88661bed129cf6fbcb59 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-9708862e1e7bf6faf85e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DBSALT001389 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Fosfestrol |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 3032325 |
|---|
| Kegg Compound ID | C08145 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|