Identification Common Name Valnemulin Class Small Molecule Description Not Available Contaminant Sources STOFF IDENT Compounds
ToxCast & Tox21 Chemicals Contaminant Type Not Available Chemical Structure Synonyms Value Source (2R)-2-Amino-N-{2-[(2-{[(1S,2R,3S,4S,6R,7R,8R,14R)-4-ethenyl-3-hydroxy-2,4,7,14-tetramethyl-9-oxotricyclo[5.4.3.0,]tetradecan-6-yl]oxy}-2-oxoethyl)sulfanyl]-2-methylpropyl}-3-methylbutanimidate Generator (2R)-2-Amino-N-{2-[(2-{[(1S,2R,3S,4S,6R,7R,8R,14R)-4-ethenyl-3-hydroxy-2,4,7,14-tetramethyl-9-oxotricyclo[5.4.3.0,]tetradecan-6-yl]oxy}-2-oxoethyl)sulphanyl]-2-methylpropyl}-3-methylbutanimidate Generator (2R)-2-Amino-N-{2-[(2-{[(1S,2R,3S,4S,6R,7R,8R,14R)-4-ethenyl-3-hydroxy-2,4,7,14-tetramethyl-9-oxotricyclo[5.4.3.0,]tetradecan-6-yl]oxy}-2-oxoethyl)sulphanyl]-2-methylpropyl}-3-methylbutanimidic acid Generator Econor MeSH
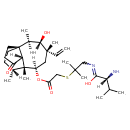
Chemical Formula C31 H52 N2 O5 S Average Molecular Mass 564.830 g/mol Monoisotopic Mass 564.360 g/mol CAS Registry Number 101312-92-9 IUPAC Name (2R)-2-amino-N-{2-[(2-{[(1S,2R,3S,4S,6R,7R,8R,14R)-4-ethenyl-3-hydroxy-2,4,7,14-tetramethyl-9-oxotricyclo[5.4.3.0¹,⁸]tetradecan-6-yl]oxy}-2-oxoethyl)sulfanyl]-2-methylpropyl}-3-methylbutanimidic acid Traditional Name valnemulin SMILES [H][C@@](N)(C(C)C)C(O)=NCC(C)(C)SCC(=O)O[C@]1([H])C[C@@](C)(C=C)[C@@]([H])(O)[C@]([H])(C)[C@]23CCC(=O)[C@@]2([H])[C@@]1(C)[C@]([H])(C)CC3 InChI Identifier InChI=1S/C31H52N2O5S/c1-10-29(8)15-22(38-23(35)16-39-28(6,7)17-33-27(37)24(32)18(2)3)30(9)19(4)11-13-31(20(5)26(29)36)14-12-21(34)25(30)31/h10,18-20,22,24-26,36H,1,11-17,32H2,2-9H3,(H,33,37)/t19-,20+,22-,24-,25+,26+,29-,30+,31+/m1/s1 InChI Key LLYYNOVSVPBRGV-MVNKZKPCSA-N