| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 05:54:23 UTC |
|---|
| Update Date | 2016-11-09 01:15:56 UTC |
|---|
| Accession Number | CHEM019199 |
|---|
| Identification |
|---|
| Common Name | 3,4-Diaminopyridine |
|---|
| Class | Small Molecule |
|---|
| Description | Amifampridine, or 3,4-diaminopyridine (3,4-DAP), is a quaternary ammonium compound that blocks presynaptic potassium channels, and subsequently prolongs the action potential and increases presynaptic calcium concentrations [A33863]. It was first discovered in Scotland in the 1970s and its clinical effectiveness for neuromuscular disorders, including Lambert–Eaton myasthenic syndrome (LEMS), has been investigated in the 1980s [L3171]. Amifampridine phosphate is a more stable salt that serves as an active ingredient of EMA-approved Firdapse, which was previously marketed as Zenas. It is currently used as the first-line symptomatic treatment for LEMS in adult patients and is ideally given as oral tablets in divided doses, three or four times a day. Firdapse (amifampridine) was formally approved by the US FDA for the treatment of adults with LEMS as recently as November of 2018 [L4819].
LEMS is a rare auto-immune disorder of the neuromuscular junction that is characterized by proximal muscle weakness, depressed tendon reflexes, and posttetanic potentiation in addition to autonomic dysfunction [A33863]. About 50-60% of the patients develop more rapidly progressive LEMS and small cell lung cancer, which influences the prognosis [A33863]. Patients with LEMS develop serum antibodies against presynaptic P/Q-type voltage-gated calcium channels, leading to decreased presynaptic calcium levels and reduced quantal release of acetylcholine, which is mainly responsible for causing symptoms of LEMS [A33863]. Reduced acetylcholine release at the neuromuscular junction leads to decreased frequency of miniature endplate potentials of normal amplitude, and insufficient acetylcholine levels for the activation of postsynaptic muscle fibers following a single nerve impulse [A33863]. This leads to the reduction of the compound muscle action potential (CMAP) [A33863]. Treatment for LEMS include immunotherapy such as conventional immunosuppression or intravenous immunoglobulins, however such treatments are recommended in patients in whom symptomatic treatment would not suffice [A33863]. Amifampridine is the nonimmune treatment options for LEMS.
In phase III clinical trials of adult patients with LEMS, treatment of amifampridine significantly improved symptoms of LEMS compared to placebo with good tolerance [A33864]. It was demonstrated in clinical studies involving healthy volunteers that the pharmacokinetics and systemic exposure to amifampridine is affected by the genetic differences in N-acetyl-transferase (NAT) enzymes (acetylator phenotype) and NAT2 genotype, which is subject to genetic variation [F272]. Slow acetylators were at higher risk for experiencing drug-associated adverse reactions, such as paresthesias, nausea, and headache [F272]. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
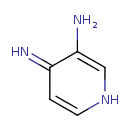
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Ruzurgi | Kegg | | Firdapse | KEGG, MeSH, HMDB | | 3,4-Diaminopyridine | MeSH | | 3,4 Diaminopyridine | MeSH, HMDB | | Amifampridine | MeSH, HMDB | | Amifampridine phosphate | MeSH, HMDB |
|
|---|
| Chemical Formula | C5H7N3 |
|---|
| Average Molecular Mass | 109.132 g/mol |
|---|
| Monoisotopic Mass | 109.064 g/mol |
|---|
| CAS Registry Number | 54-96-6 |
|---|
| IUPAC Name | 4-imino-1,4-dihydropyridin-3-amine |
|---|
| Traditional Name | 3,4-diaminopyridine |
|---|
| SMILES | NC1=CNC=CC1=N |
|---|
| InChI Identifier | InChI=1S/C5H7N3/c6-4-1-2-8-3-5(4)7/h1-3H,7H2,(H2,6,8) |
|---|
| InChI Key | OYTKINVCDFNREN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as aminopyridines and derivatives. These are organic heterocyclic compounds containing an amino group attached to a pyridine ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyridines and derivatives |
|---|
| Sub Class | Aminopyridines and derivatives |
|---|
| Direct Parent | Aminopyridines and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aminopyridine
- Heteroaromatic compound
- Azacycle
- Organic nitrogen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Primary amine
- Organonitrogen compound
- Amine
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0a4i-9500000000-a63c0bd9a1bb53b616fd | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-7352311900-1c848af532bb5f96c1d6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0900000000-26fff8629faf8b47107f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-2900000000-aeca16edcaa5f9640041 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-9000000000-a5af7b664986d05c3634 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0900000000-ff94645e19722e1e32e4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0900000000-4fffbabe99bdd1adfa0d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9600000000-d415f9f018514ffedcdb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0900000000-098c82e0975dbfe9a0ea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-2900000000-4ac722d417c259b9b90e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udl-9000000000-b26dd6b9df02cc775841 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0900000000-11720ab9074f1da461a6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-1900000000-b18c16381a318d30b60a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-4900000000-e58ed6c87021dce87f7c | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB11640 |
|---|
| HMDB ID | HMDB0304888 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 5705 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|