Identification Common Name Eptifibatide Class Small Molecule Description A synthetic homodetic cyclic peptide comprising N(alpha)-(3-sulfanylpropanoyl)homoarginyl, glycyl, aspartyl, tryptophyl, prolyl and cysteinamide residues connected in sequence and cyclised via a disulfide bond. Derived from a protein found in the venom of the southeastern pygmy rattlesnake, Sistrurus miliarus barbouri, eptifibatide is an anti-coagulant that inhibits platelet aggregation by selectively blocking the platelet glycoprotein IIb/IIIa receptor, so preventing the binding of fibrinogen, von Willebrand factor, and other adhesive ligands. It is used in the management of unstable angina and in patients undergoing coronary angioplasty and stenting procedures. Contaminant Sources STOFF IDENT Compounds
ToxCast & Tox21 Chemicals Contaminant Type Not Available Chemical Structure Synonyms Value Source N(6)-Amidino-N(2)-(3-mercaptopropionyl)-L-lysylglycyl-L-alpha-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic(1-6)-disulfide ChEBI S(1),S(6)-Cyclo[N(6)-carbamimidoyl-N(2)-(3-sulfanylpropanoyl)-L-lysylglycyl-L-alpha-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide] ChEBI N(6)-Amidino-N(2)-(3-mercaptopropionyl)-L-lysylglycyl-L-a-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic(1-6)-disulfide Generator N(6)-Amidino-N(2)-(3-mercaptopropionyl)-L-lysylglycyl-L-a-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic(1-6)-disulphide Generator N(6)-Amidino-N(2)-(3-mercaptopropionyl)-L-lysylglycyl-L-alpha-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic(1-6)-disulphide Generator N(6)-Amidino-N(2)-(3-mercaptopropionyl)-L-lysylglycyl-L-α-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic(1-6)-disulfide Generator N(6)-Amidino-N(2)-(3-mercaptopropionyl)-L-lysylglycyl-L-α-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic(1-6)-disulphide Generator S(1),S(6)-Cyclo[N(6)-carbamimidoyl-N(2)-(3-sulfanylpropanoyl)-L-lysylglycyl-L-a-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide] Generator S(1),S(6)-Cyclo[N(6)-carbamimidoyl-N(2)-(3-sulfanylpropanoyl)-L-lysylglycyl-L-α-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide] Generator S(1),S(6)-Cyclo[N(6)-carbamimidoyl-N(2)-(3-sulphanylpropanoyl)-L-lysylglycyl-L-a-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide] Generator S(1),S(6)-Cyclo[N(6)-carbamimidoyl-N(2)-(3-sulphanylpropanoyl)-L-lysylglycyl-L-alpha-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide] Generator S(1),S(6)-Cyclo[N(6)-carbamimidoyl-N(2)-(3-sulphanylpropanoyl)-L-lysylglycyl-L-α-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide] Generator Integrelin MeSH Integrilin MeSH Epifibatide MeSH Epifibratide MeSH
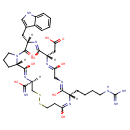
Chemical Formula C35 H49 N11 O9 S2 Average Molecular Mass 831.970 g/mol Monoisotopic Mass 831.316 g/mol CAS Registry Number 188627-80-7 IUPAC Name 2-[(3R,11S,17S,20S,25aS)-11-(4-carbamimidamidobutyl)-1,9,12,15,18-pentahydroxy-3-(C-hydroxycarbonimidoyl)-20-[(1H-indol-3-yl)methyl]-21-oxo-3H,4H,7H,8H,11H,14H,17H,20H,21H,23H,24H,25H,25aH-pyrrolo[2,1-g]1,2-dithia-5,8,11,14,17,20-hexaazacyclotricosan-17-yl]acetic acid Traditional Name eptifibatide SMILES [H][C@@]12CCCN1C(=O)[C@]([H])(CC1=CNC3=CC=CC=C13)N=C(O)[C@]([H])(CC(O)=O)N=C(O)CN=C(O)[C@]([H])(CCCCNC(N)=N)N=C(O)CCSSC[C@]([H])(N=C2O)C(O)=N InChI Identifier InChI=1S/C35H49N11O9S2/c36-30(51)25-18-57-56-13-10-27(47)42-22(8-3-4-11-39-35(37)38)31(52)41-17-28(48)43-23(15-29(49)50)32(53)44-24(14-19-16-40-21-7-2-1-6-20(19)21)34(55)46-12-5-9-26(46)33(54)45-25/h1-2,6-7,16,22-26,40H,3-5,8-15,17-18H2,(H2,36,51)(H,41,52)(H,42,47)(H,43,48)(H,44,53)(H,45,54)(H,49,50)(H4,37,38,39)/t22-,23-,24-,25-,26-/m0/s1 InChI Key CZKPOZZJODAYPZ-LROMGURASA-N Chemical Taxonomy Description belongs to the class of organic compounds known as glutamic acid and derivatives. Glutamic acid and derivatives are compounds containing glutamic acid or a derivative thereof resulting from reaction of glutamic acid at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. Kingdom Organic compounds Super Class Organic acids and derivatives Class Carboxylic acids and derivatives Sub Class Amino acids, peptides, and analogues Direct Parent Glutamic acid and derivatives Alternative Parents Substituents Glutamic acid or derivatives
N-acyl-alpha amino acid or derivatives
N-acyl-alpha-amino acid
Hippuric acid
Hippuric acid or derivatives
Benzamide
Pyrrolo[2,3-d]pyrimidine
Benzoic acid or derivatives
Pyrrolopyrimidine
Benzoyl
Aminopyrimidine
Pyrimidone
Monocyclic benzene moiety
Pyrimidine
Substituted pyrrole
Benzenoid
Dicarboxylic acid or derivatives
Pyrrole
Vinylogous amide
Heteroaromatic compound
Amino acid
Secondary carboxylic acid amide
Carboxamide group
Carboxylic acid salt
Azacycle
Organic alkali metal salt
Carboxylic acid
Organoheterocyclic compound
Organic salt
Organic oxygen compound
Organic sodium salt
Organic nitrogen compound
Amine
Carbonyl group
Hydrocarbon derivative
Organic oxide
Organopnictogen compound
Organic zwitterion
Primary amine
Organonitrogen compound
Organooxygen compound
Aromatic heteropolycyclic compound Molecular Framework Aromatic heteropolycyclic compounds External Descriptors