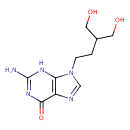
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-024r-4890000000-ed3881f9b07a40fef086 | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-0089-4039000000-39d85efcb22394abeb78 | Spectrum |
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , negative | splash10-0udi-0190000000-ed1701b795cb216e6d66 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , negative | splash10-0udi-0790000000-212e25de28163f343cac | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , negative | splash10-0udi-0910000000-3a8ca3a0b3701c27b51d | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , negative | splash10-0kai-0900000000-36122e670b73f1113fff | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , negative | splash10-053r-1900000000-bceccffce9861527c0c8 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , negative | splash10-0api-3900000000-2322dab886ffdf508841 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0udi-0490000000-0e41d127a3f6d343b9d6 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0udi-0920000000-8e58b381a6b3d470746b | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0udi-0900000000-d498bce256e98e4cf88c | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0udi-0900000000-ae0beed2651e1125764f | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0udi-0900000000-19ad8b8788d0275996b5 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0udr-1900000000-5c4ec47a69c82954d428 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0f79-0390000000-2bec5fbdf1e72aa84bde | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0uei-1940000000-6e49677068b47b74940f | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0udi-3900000000-cc1f9ce4e3ed563ded5c | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0zfr-8900000000-326741cdbb592bb9d271 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0f8c-7900000000-295551b253f3f85729c3 | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-IT , positive | splash10-0udi-0900000000-44a1abcea55a53906b2a | Spectrum |
| LC-MS/MS | LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0980000000-aaaff73f565024ce9cff | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udr-0090000000-b771df12ed0fcbbee1ca | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udr-1980000000-be68269fbcfeb1eda4f2 | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udr-4900000000-54f7176cadc1b67a66dc | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0090000000-b42cd4e9561cb4baa03a | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0960000000-012e01c533768b510f86 | Spectrum |
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-4900000000-8dda187de53e2d7eb1fb | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
| 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |