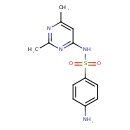
| (p-Aminobenzolsulfonyl)-4-amino-2,6-dimethylpyrimidin | ChEBI |
| (p-Aminobenzolsulfonyl)-4-amino-2,6-dimethylpyrimidine | ChEBI |
| 2,4-Dimethyl-6-sulfanilamidopyrimidine | ChEBI |
| 2,6-Dimethyl-4-sulfanilamidopyrimidine | ChEBI |
| 4-Sulfa-2,6-dimethylpyrimidine | ChEBI |
| 4-Sulfanilamido-2,6-dimethylpyrimidine | ChEBI |
| 6-(4-Aminobenzenesulfonamido)-2,4-dimethylpyrimidine | ChEBI |
| 6-(p-Aminobenzenesulfonamido)-2,4-dimethylpyrimidine | ChEBI |
| 6-(p-Aminobenzenesulfonyl)amino-2,4-dimethylpyrimidine | ChEBI |
| 6-Sulfanilamido-2,4-dimethylpyrimidine | ChEBI |
| N(1)-(2,6-Dimethyl-4-pyrimidinyl)sulfanilamide | ChEBI |
| Solfisomidina | ChEBI |
| Sulfaisodimidinum | ChEBI |
| Sulfasomidine | ChEBI |
| Sulphasomidine | ChEBI |
| (p-Aminobenzolsulphonyl)-4-amino-2,6-dimethylpyrimidin | Generator |
| (p-Aminobenzolsulphonyl)-4-amino-2,6-dimethylpyrimidine | Generator |
| 2,4-Dimethyl-6-sulphanilamidopyrimidine | Generator |
| 2,6-Dimethyl-4-sulphanilamidopyrimidine | Generator |
| 4-Sulpha-2,6-dimethylpyrimidine | Generator |
| 4-Sulphanilamido-2,6-dimethylpyrimidine | Generator |
| 6-(4-Aminobenzenesulphonamido)-2,4-dimethylpyrimidine | Generator |
| 6-(p-Aminobenzenesulphonamido)-2,4-dimethylpyrimidine | Generator |
| 6-(p-Aminobenzenesulphonyl)amino-2,4-dimethylpyrimidine | Generator |
| 6-Sulphanilamido-2,4-dimethylpyrimidine | Generator |
| N(1)-(2,6-Dimethyl-4-pyrimidinyl)sulphanilamide | Generator |
| Sulphaisodimidinum | Generator |
| Sulphaisodimidine | Generator |
| Sulfaisodimidine | ChEBI |
| Sulphisomidine | Generator |
| Augensalbe, aristamid | MeSH |
| Elkosin | MeSH |
| Optima brand OF sulfisomidine | MeSH |
| Aristamid augentropfen | MeSH |
| Aristamid augensalbe | MeSH |
| Sulfisomidine sodium | MeSH |
| Augentropfen, aristamid | MeSH |
| Optima brand OF sulfisomidine sodium | MeSH |
| Sodium, sulfisomidine | MeSH |