| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 05:32:30 UTC |
|---|
| Update Date | 2016-11-09 01:15:52 UTC |
|---|
| Accession Number | CHEM018798 |
|---|
| Identification |
|---|
| Common Name | Crocin |
|---|
| Class | Small Molecule |
|---|
| Description | A diester that is crocetin in which both of the carboxy groups have been converted to their gentiobiosyl esters. It is one of the water-soluble yellow-red pigments of saffron and is used as a spice for flavouring and colouring food. Note that in India, the term 'Crocin' is also used by GlaxoSmithKline as a brand-name for paracetamol. |
|---|
| Contaminant Sources | - FooDB Chemicals
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
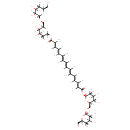
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| alpha-Crocin | ChEBI | | Bis(beta-D-gentiobiosyl) crocetin | ChEBI | | C.I. 75100 | ChEBI | | Crocetin di-beta-D-gentiobiose ester | ChEBI | | Crocetin di-gentiobiose ester | ChEBI | | Crocetin digentiobioside | ChEBI | | Crocine | ChEBI | | Natural red 1 | ChEBI | | Natural yellow 19 | ChEBI | | Natural yellow 6 | ChEBI | | Saffron | ChEBI | | trans-Crocetin bis(beta-D-gentiobiosyl) ester | ChEBI | | a-Crocin | Generator | | Α-crocin | Generator | | Bis(b-D-gentiobiosyl) crocetin | Generator | | Bis(β-D-gentiobiosyl) crocetin | Generator | | Crocetin di-b-D-gentiobiose ester | Generator | | Crocetin di-β-D-gentiobiose ester | Generator | | trans-Crocetin bis(b-D-gentiobiosyl) ester | Generator | | trans-Crocetin bis(β-D-gentiobiosyl) ester | Generator | | all-trans-Crocetin di-beta-D-gentiobiosyl ester | HMDB | | all-trans-Crocetin di-beta-delta-gentiobiosyl ester | HMDB | | Crocetin bis(gentiobiosyl) ester | HMDB | | Crocetin digentiobiose ester | HMDB | | Crocin 1 | HMDB | | Gardenia yellow | HMDB | | Saffron (JP15) | HMDB | | Crocin | MeSH |
|
|---|
| Chemical Formula | C44H64O24 |
|---|
| Average Molecular Mass | 976.972 g/mol |
|---|
| Monoisotopic Mass | 976.379 g/mol |
|---|
| CAS Registry Number | 42553-65-1 |
|---|
| IUPAC Name | bis[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-({[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}methyl)oxan-2-yl] (2E,4E,6E,8E,10E,12E,14E)-2,6,11,15-tetramethylhexadeca-2,4,6,8,10,12,14-heptaenedioate |
|---|
| Traditional Name | saffron |
|---|
| SMILES | C\C(\C=C\C=C(/C)C(=O)O[C@@H]1O[C@H](CO[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)[C@@H](O)[C@H](O)[C@H]1O)=C/C=C/C=C(\C)/C=C/C=C(\C)C(=O)O[C@@H]1O[C@H](CO[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)[C@@H](O)[C@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C44H64O24/c1-19(11-7-13-21(3)39(59)67-43-37(57)33(53)29(49)25(65-43)17-61-41-35(55)31(51)27(47)23(15-45)63-41)9-5-6-10-20(2)12-8-14-22(4)40(60)68-44-38(58)34(54)30(50)26(66-44)18-62-42-36(56)32(52)28(48)24(16-46)64-42/h5-14,23-38,41-58H,15-18H2,1-4H3/b6-5+,11-7+,12-8+,19-9+,20-10+,21-13+,22-14+/t23-,24-,25-,26-,27-,28-,29-,30-,31+,32+,33+,34+,35-,36-,37-,38-,41-,42-,43+,44+/m1/s1 |
|---|
| InChI Key | SEBIKDIMAPSUBY-RTJKDTQDSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as diterpenoids. These are terpene compounds formed by four isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | Diterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diterpenoid
- Disaccharide
- Glycosyl compound
- O-glycosyl compound
- Fatty acid ester
- Dicarboxylic acid or derivatives
- Fatty acyl
- Oxane
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- Carboxylic acid ester
- Secondary alcohol
- Polyol
- Acetal
- Organoheterocyclic compound
- Oxacycle
- Carboxylic acid derivative
- Alcohol
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Primary alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 10V, Negative | splash10-0fb9-0000008009-3b75a0bb1954441908e5 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0002009000-9e622eb534e813be910c | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 40V, Negative | splash10-0059-1259001000-27e1be6013a6ff14a567 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0570-0107109005-8d4ac227319c8db4f8a8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01p9-0316529102-33fcee1955a03eb2acbe | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03mr-1948544002-585ea2a4ab28ee86f4ac | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-1514037019-77be269ba2dd85c94f6b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0c29-3914005115-42287fc2024b10a75c14 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0uml-8933015221-e452e968e696d68b4e6f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a70-0101229114-188a76b2b8dade56be41 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ce9-0384029001-234bd0906e1d9e993e0b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-0491033013-b4cd69b33ee6311f8a36 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0pk9-0018019003-dec192fc122debfcf133 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0bt9-4200019082-19b8afa05b75bf20b85b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-2000009001-de243920ac2be1b58304 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB11874 |
|---|
| HMDB ID | HMDB0002398 |
|---|
| FooDB ID | FDB014549 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00003769 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | CPD-8666 |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Crocin |
|---|
| Chemspider ID | 4444645 |
|---|
| ChEBI ID | 79068 |
|---|
| PubChem Compound ID | 5281233 |
|---|
| Kegg Compound ID | C08589 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. https://www.ncbi.nlm.nih.gov/pubmed/?term=14514073 | | 2. https://www.ncbi.nlm.nih.gov/pubmed/?term=17704979 | | 3. https://www.ncbi.nlm.nih.gov/pubmed/?term=21398065 | | 4. https://www.ncbi.nlm.nih.gov/pubmed/?term=24132704 | | 5. https://www.ncbi.nlm.nih.gov/pubmed/?term=24275090 | | 6. https://www.ncbi.nlm.nih.gov/pubmed/?term=24401376 | | 7. https://www.ncbi.nlm.nih.gov/pubmed/?term=24659065 | | 8. https://www.ncbi.nlm.nih.gov/pubmed/?term=24697694 | | 9. https://www.ncbi.nlm.nih.gov/pubmed/?term=24839356 | | 10. Ochiai T, Shimeno H, Mishima K, Iwasaki K, Fujiwara M, Tanaka H, Shoyama Y, Toda A, Eyanagi R, Soeda S: Protective effects of carotenoids from saffron on neuronal injury in vitro and in vivo. Biochim Biophys Acta. 2007 Apr;1770(4):578-84. Epub 2006 Dec 5. | | 11. Carmona M, Zalacain A, Sanchez AM, Novella JL, Alonso GL: Crocetin esters, picrocrocin and its related compounds present in Crocus sativus stigmas and Gardenia jasminoides fruits. Tentative identification of seven new compounds by LC-ESI-MS. J Agric Food Chem. 2006 Feb 8;54(3):973-9. |
|
|---|