Identification Common Name Tilmicosin Class Small Molecule Description Tilmicosin is a macrolide antibiotic. It is used in veterinary medicine for the treatment of bovine respiratory disease and ovine respiratory disease associated with _Mannheimia haemolytica_. Contaminant Sources STOFF IDENT Compounds
Suspected Compounds – Schymanski Project
ToxCast & Tox21 Chemicals Contaminant Type Not Available Chemical Structure Synonyms Value Source TMS Kegg Micotil Kegg 20-deoxo-20-(3,5-Dimethylpiperidin-1-yl)desmycosin MeSH MICOTIL 300 MeSH Tilmicosin MeSH
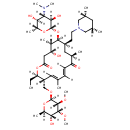
Chemical Formula C46 H80 N2 O13 Average Molecular Mass 869.147 g/mol Monoisotopic Mass 868.566 g/mol CAS Registry Number 108050-54-0 IUPAC Name (4R,5S,6S,7R,9R,11E,13E,15R,16R)-6-{[(2R,3R,4S,5S,6R)-4-(dimethylamino)-3,5-dihydroxy-6-methyloxan-2-yl]oxy}-7-{2-[(3R,5S)-3,5-dimethylpiperidin-1-yl]ethyl}-16-ethyl-4-hydroxy-15-({[(2R,3R,4R,5R,6R)-5-hydroxy-3,4-dimethoxy-6-methyloxan-2-yl]oxy}methyl)-5,9,13-trimethyl-1-oxacyclohexadeca-11,13-diene-2,10-dione Traditional Name tilmicosin SMILES [H]\C1=C([H])/C(=O)[C@]([H])(C)C[C@]([H])(CCN2C[C@]([H])(C)C[C@]([H])(C)C2)[C@]([H])(O[C@]2([H])O[C@]([H])(C)[C@@]([H])(O)[C@]([H])(N(C)C)[C@@]2([H])O)[C@@]([H])(C)[C@]([H])(O)CC(=O)O[C@]([H])(CC)[C@@]([H])(CO[C@]2([H])O[C@]([H])(C)[C@@]([H])(O)[C@@]([H])(OC)[C@@]2([H])OC)C([H])=C1C InChI Identifier InChI=1S/C46H80N2O13/c1-13-36-33(24-57-46-44(56-12)43(55-11)40(53)31(8)59-46)19-25(2)14-15-34(49)28(5)20-32(16-17-48-22-26(3)18-27(4)23-48)42(29(6)35(50)21-37(51)60-36)61-45-41(54)38(47(9)10)39(52)30(7)58-45/h14-15,19,26-33,35-36,38-46,50,52-54H,13,16-18,20-24H2,1-12H3/b15-14+,25-19+/t26-,27+,28-,29+,30-,31-,32+,33-,35-,36-,38+,39-,40-,41-,42-,43-,44-,45+,46-/m1/s1 InChI Key JTSDBFGMPLKDCD-XVFHVFLVSA-N Chemical Taxonomy Description belongs to the class of organic compounds known as imidazole ribonucleosides and ribonucleotides. These are organic compounds in which the C-1 of a ribosyl moiety is N-linked to an imidazole ring. Nucleotides have a phosphate group linked to the C5 carbon of the ribose (or deoxyribose) moiety. This class does not contain benzimidazole nucleosides and nucleotides. Kingdom Organic compounds Super Class Nucleosides, nucleotides, and analogues Class Imidazole ribonucleosides and ribonucleotides Sub Class Not Available Direct Parent Imidazole ribonucleosides and ribonucleotides Alternative Parents Substituents Imidazole ribonucleoside
Glycosyl compound
N-glycosyl compound
Pentose monosaccharide
Aminoimidazole
Monosaccharide
N-substituted imidazole
Azole
Heteroaromatic compound
Imidazole
Tetrahydrofuran
Secondary alcohol
Carboximidic acid
Carboximidic acid derivative
Oxacycle
Azacycle
Organoheterocyclic compound
Organic nitrogen compound
Hydrocarbon derivative
Organopnictogen compound
Amine
Alcohol
Organic oxygen compound
Primary amine
Organonitrogen compound
Organooxygen compound
Primary alcohol
Aromatic heteromonocyclic compound Molecular Framework Aromatic heteromonocyclic compounds External Descriptors