| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 05:24:47 UTC |
|---|
| Update Date | 2016-11-09 01:15:50 UTC |
|---|
| Accession Number | CHEM018637 |
|---|
| Identification |
|---|
| Common Name | Clobetasol propionate |
|---|
| Class | Small Molecule |
|---|
| Description | Clobetasol propionate is a prednisolone derivative with higher specificity for glucocorticoid receptors than mineralocorticoid receptors.[L11815] It has demonstrated superior activity compared to [fluocinonide][A190963] and was first described in the literature in 1974.[A190936]
Clobetasol Propionate was granted FDA approval on 27 December 1985.[L11809] |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
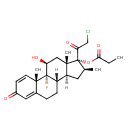
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 21-Chloro-9-fluoro-11beta,17-dihydroxy-16beta-methylpregna-1,4-diene-3,20-dione 17-propionate | ChEBI | | Clobetasol 17-propanoate | ChEBI | | Clobetasol 17-propionate | ChEBI | | Clobex | Kegg | | Cormax | Kegg | | Olux | Kegg | | 21-Chloro-9-fluoro-11b,17-dihydroxy-16b-methylpregna-1,4-diene-3,20-dione 17-propionate | Generator | | 21-Chloro-9-fluoro-11b,17-dihydroxy-16b-methylpregna-1,4-diene-3,20-dione 17-propionic acid | Generator | | 21-Chloro-9-fluoro-11beta,17-dihydroxy-16beta-methylpregna-1,4-diene-3,20-dione 17-propionic acid | Generator | | 21-Chloro-9-fluoro-11β,17-dihydroxy-16β-methylpregna-1,4-diene-3,20-dione 17-propionate | Generator | | 21-Chloro-9-fluoro-11β,17-dihydroxy-16β-methylpregna-1,4-diene-3,20-dione 17-propionic acid | Generator | | Clobetasol 17-propanoic acid | Generator | | Clobetasol 17-propionic acid | Generator | | Clobetasol propionic acid | Generator | | Embeline e | MeSH | | Clobetasol 17 propionate | MeSH | | Clofenazon | MeSH | | Embeline | MeSH | | Temovate | MeSH | | Clobetasol | MeSH | | Dermovate | MeSH |
|
|---|
| Chemical Formula | C25H32ClFO5 |
|---|
| Average Molecular Mass | 466.970 g/mol |
|---|
| Monoisotopic Mass | 466.192 g/mol |
|---|
| CAS Registry Number | 25122-46-7 |
|---|
| IUPAC Name | (1R,2S,10S,11S,13S,14R,15S,17S)-14-(2-chloroacetyl)-1-fluoro-17-hydroxy-2,13,15-trimethyl-5-oxotetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadeca-3,6-dien-14-yl propanoate |
|---|
| Traditional Name | temovate |
|---|
| SMILES | [H][C@@]12C[C@H](C)[C@](OC(=O)CC)(C(=O)CCl)[C@@]1(C)C[C@H](O)[C@@]1(F)[C@@]2([H])CCC2=CC(=O)C=C[C@]12C |
|---|
| InChI Identifier | InChI=1S/C25H32ClFO5/c1-5-21(31)32-25(20(30)13-26)14(2)10-18-17-7-6-15-11-16(28)8-9-22(15,3)24(17,27)19(29)12-23(18,25)4/h8-9,11,14,17-19,29H,5-7,10,12-13H2,1-4H3/t14-,17-,18-,19-,22-,23-,24-,25-/m0/s1 |
|---|
| InChI Key | CBGUOGMQLZIXBE-XGQKBEPLSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as gluco/mineralocorticoids, progestogins and derivatives. These are steroids with a structure based on a hydroxylated prostane moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Pregnane steroids |
|---|
| Direct Parent | Gluco/mineralocorticoids, progestogins and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Progestogin-skeleton
- Steroid ester
- 20-oxosteroid
- 3-oxo-delta-1,4-steroid
- 3-oxosteroid
- 11-hydroxysteroid
- 11-beta-hydroxysteroid
- Oxosteroid
- 9-halo-steroid
- Halo-steroid
- Hydroxysteroid
- Delta-1,4-steroid
- Alpha-acyloxy ketone
- Alpha-haloketone
- Alpha-chloroketone
- Cyclic alcohol
- Carboxylic acid ester
- Cyclic ketone
- Fluorohydrin
- Secondary alcohol
- Halohydrin
- Ketone
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Organooxygen compound
- Alcohol
- Organic oxide
- Hydrocarbon derivative
- Organic oxygen compound
- Carbonyl group
- Alkyl halide
- Organofluoride
- Alkyl chloride
- Organochloride
- Alkyl fluoride
- Organohalogen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-05fr-1920000000-defac8a82087405b74df | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-0a6r-0596000000-b19d97ac39952d18b68e | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-03mj-3971000000-8f42562c23aaf5e5d240 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-066s-3003900000-de1fe5b252dd99c47da6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-9036600000-8d6e9893a1d817fa1237 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a59-4298100000-9d10026ac5aee897f91f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-2002900000-3d0b3c5111e9b7289d57 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ab9-5009800000-c98c0202af4a862421a0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ab9-8009000000-66051bd53af18b72b922 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01013 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Clobetasol |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 31414 |
|---|
| PubChem Compound ID | 32798 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|