| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 05:18:03 UTC |
|---|
| Update Date | 2016-11-09 01:15:49 UTC |
|---|
| Accession Number | CHEM018509 |
|---|
| Identification |
|---|
| Common Name | Pyricarbate |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
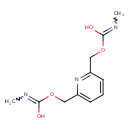
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Pyricarbate | Kegg | | Anginin | Kegg | | Angioxine | Kegg | | Pyridinol carbamate | Kegg | | Pyricarbic acid | Generator | | Pyridinol carbamic acid | Generator | | N-Methyl[(6-{[(methyl-C-hydroxycarbonimidoyl)oxy]methyl}pyridin-2-yl)methoxy]carboximidate | Generator | | Parmidine | MeSH | | Duvaline | MeSH | | Prodectin | MeSH | | Pyridinolcarbamate | MeSH | | Lisator | MeSH | | Colesterinex | MeSH | | Gasparol | MeSH | | Movecil | MeSH | | Sospitan | MeSH | | 2,6-Pyridinedimethanol-bis-(N-methylcarbamate) | MeSH | | Duaxol | MeSH |
|
|---|
| Chemical Formula | C11H15N3O4 |
|---|
| Average Molecular Mass | 253.258 g/mol |
|---|
| Monoisotopic Mass | 253.106 g/mol |
|---|
| CAS Registry Number | 1882-26-4 |
|---|
| IUPAC Name | N-methyl[(6-{[(methyl-C-hydroxycarbonimidoyl)oxy]methyl}pyridin-2-yl)methoxy]carboximidic acid |
|---|
| Traditional Name | anginine |
|---|
| SMILES | CN=C(O)OCC1=CC=CC(COC(O)=NC)=N1 |
|---|
| InChI Identifier | InChI=1S/C11H15N3O4/c1-12-10(15)17-6-8-4-3-5-9(14-8)7-18-11(16)13-2/h3-5H,6-7H2,1-2H3,(H,12,15)(H,13,16) |
|---|
| InChI Key | YEKQSSHBERGOJK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pyridines and derivatives. Pyridines and derivatives are compounds containing a pyridine ring, which is a six-member aromatic heterocycle which consists of one nitrogen atom and five carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyridines and derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Pyridines and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pyridine
- Heteroaromatic compound
- Azacycle
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Carboximidic acid derivative
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Imine
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-057i-6910000000-9c3334b255ac34ac3ce1 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ufr-7390000000-10937648ac66639c9b17 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a6r-9820000000-2631df9de0716fab92c1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-9200000000-24cfdfe482a9b2cb7f0a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9120000000-8b3972a69d31225ef9ae | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9110000000-cd1000bbfaa9ccf7374f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9100000000-cefc9ab57fc0f9b8b04d | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0256959 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4816 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | C12842 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|