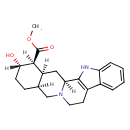
| 17alpha-Hydroxy-20alpha-yohimban-16beta-carboxylic acid methyl ester | ChEBI |
| alpha-Yohimbine | ChEBI |
| Corynanthidine | ChEBI |
| Isoyohimbine | ChEBI |
| Mesoyohimbine | ChEBI |
| Methyl (16beta,17alpha,20alpha)-17-hydroxyyohimban-16-carboxylate | ChEBI |
| 17a-Hydroxy-20a-yohimban-16b-carboxylate methyl ester | Generator |
| 17a-Hydroxy-20a-yohimban-16b-carboxylic acid methyl ester | Generator |
| 17alpha-Hydroxy-20alpha-yohimban-16beta-carboxylate methyl ester | Generator |
| 17Α-hydroxy-20α-yohimban-16β-carboxylate methyl ester | Generator |
| 17Α-hydroxy-20α-yohimban-16β-carboxylic acid methyl ester | Generator |
| a-Yohimbine | Generator |
| Α-yohimbine | Generator |
| Methyl (16b,17a,20a)-17-hydroxyyohimban-16-carboxylate | Generator |
| Methyl (16b,17a,20a)-17-hydroxyyohimban-16-carboxylic acid | Generator |
| Methyl (16beta,17alpha,20alpha)-17-hydroxyyohimban-16-carboxylic acid | Generator |
| Methyl (16β,17α,20α)-17-hydroxyyohimban-16-carboxylate | Generator |
| Methyl (16β,17α,20α)-17-hydroxyyohimban-16-carboxylic acid | Generator |
| Methyl (1S,15S,18S,19S,20S)-18-hydroxy-1,3,11,12,14,15,16,17,18,19,20,21-dodecahydroyohimban-19-carboxylic acid | Generator |
| Aphrodyne | MeSH |
| Hydrochloride, aphrodine | MeSH |
| Solvay brand OF yohimbine hydrochloride | MeSH |
| Yocon | MeSH |
| Yohimbine | MeSH |
| Aphrodine hydrochloride | MeSH |
| Palisades brand OF yohimbine hydrochloride | MeSH |
| Pluriviron | MeSH |
| Rauwolscine | MeSH |
| Corynanthine | MeSH |
| Kramer brand OF yohimbine hydrochloride | MeSH |
| Rauhimbine | MeSH |
| Yohimbine houdé | MeSH |
| Yohimex | MeSH |
| Hydrochloride, yohimbine | MeSH |
| Yohimbin spiegel | MeSH |
| Corynanthine tartrate | MeSH |
| Glenwood brand OF yohimbine hydrochloride | MeSH |
| Star brand OF yohimbine hydrochloride | MeSH |
| Tartrate, corynanthine | MeSH |
| StegroPharm brand OF yohimbine hydrochloride | MeSH |
| Aventis brand OF yohimbine hydrochloride | MeSH |
| Yohimbine hydrochloride | MeSH |