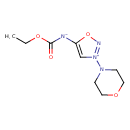
| 5-[(Ethoxycarbonyl)amino]-3-(4-morpholinyl)-1,2,3-oxadiazolium inner salt | ChEBI |
| CAS 276 | ChEBI |
| CAS-276 | ChEBI |
| Corraton | ChEBI |
| Coruno | ChEBI |
| Corvaton | ChEBI |
| Covarsal | ChEBI |
| MolsiCare | ChEBI |
| Molsicor | ChEBI |
| Molsidolat | ChEBI |
| Molsidomin | ChEBI |
| Molsidomina | ChEBI |
| Molsidominum | ChEBI |
| Morial | ChEBI |
| Morsydomine | ChEBI |
| Motazomin | ChEBI |
| N-(Ethoxycarbonyl)-3-morpholinosydnone imine | ChEBI |
| N-Carboxy-3-morpholinosydnone imine ethyl ester | ChEBI |
| N-Ethoxycarbonyl-3-morpholinosydnonimine | ChEBI |
| SIN-10 | ChEBI |
| Sydnopharm | ChEBI |
| 1-Ethoxy-N-(3-morpholin-4-yloxadiazol-3-ium-5-yl)methanimidic acid | Generator |
| Molsidain | HMDB |
| SIN 10 | HMDB |
| Molsidomin stada | HMDB |
| Von CT, molsidomin | HMDB |
| Molsi 1a pharma | HMDB |
| Molsibeta | HMDB |
| SIN10 | HMDB |
| Molsi-azu | HMDB |
| Fali cor | HMDB |
| Molsihexal | HMDB |
| Molsi azu | HMDB |
| Molsidomin ratiopharm | HMDB |
| Fali-cor | HMDB |
| Korvatone | HMDB |
| Molsidomin heumann | HMDB |
| Molsidomin-ratiopharm | HMDB |
| Molsi puren | HMDB |
| Duracoron | HMDB |
| MTW-Molsidomin | HMDB |
| Heumann, molsidomin | HMDB |
| Molsiket | HMDB |
| MTW Molsidomin | HMDB |
| Molsidomin von CT | HMDB |
| Corpea | HMDB |
| Molsi-puren | HMDB |
| Alpharma brand OF molsidomine | HMDB |
| Molsidomine normon brand | HMDB |
| Molsidominratiopharm | HMDB |
| Stadapharm brand OF molsidomine | HMDB |
| CT Arzneimittel brand OF molsidomine | HMDB |
| 1a Brand OF molsidomine | HMDB |
| Azupharma brand OF molsidomine | HMDB |
| Molsidomine ratiopharm brand | HMDB |
| Normon brand OF molsidomine | HMDB |
| Schwarz brand OF molsidomine | HMDB |
| Molsidomine | HMDB |
| Molsidomine aventis brand | HMDB |
| Molsidomine salutas brand | HMDB |
| Molsidomine schwarz brand | HMDB |
| Molsidomine CT-arzneimittel brand | HMDB |
| Sanol brand OF molsidomine | HMDB |
| CT-Arzneimittel brand OF molsidomine | HMDB |
| Hexal brand OF molsidomine | HMDB |
| Merck dura brand OF molsidomine | HMDB |
| Molsidomine heumann brand | HMDB |
| Molsidomine stadapharm brand | HMDB |
| Salutas brand OF molsidomine | HMDB |
| Heumann brand OF molsidomine | HMDB |
| Molsidomine betapharm brand | HMDB |
| Aventis brand OF molsidomine | HMDB |
| Molsidomine azupharma brand | HMDB |
| Molsidomine hexal brand | HMDB |
| Molsidomine MTW brand | HMDB |
| Betapharm brand OF molsidomine | HMDB |
| Ratiopharm brand OF molsidomine | HMDB |
| MTW Brand OF molsidomine | HMDB |
| Molsidomine 1a brand | HMDB |
| Molsidomine alpharma brand | HMDB |
| Molsidomine sanol brand | HMDB |
| Ethyl N-[3-(morpholin-4-yl)-5H-1,2,3λ⁵-oxadiazol-5-ylidene]carbamic acid | HMDB |