| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 04:41:22 UTC |
|---|
| Update Date | 2016-11-09 01:15:41 UTC |
|---|
| Accession Number | CHEM017845 |
|---|
| Identification |
|---|
| Common Name | Artesunate |
|---|
| Class | Small Molecule |
|---|
| Description | Artesunate (AS) is a medication used to treat malaria. The intravenous form is preferred to quinidine for severe malaria. Often it is used as part of combination therapy, such as artesunate plus mefloquine. It is not used for the prevention of malaria. Artesunate can be given by injection into a vein, injection into a muscle, by mouth, and by rectum.Artesunate is generally well tolerated. Side effects may include a slow heartbeat, allergic reaction, dizziness, and low white blood cell levels. During pregnancy it appears to be a safer option, even though animal studies have found harm to the baby. Use is likely okay during breastfeeding. It is in the artemisinin class of medication.Artesunate is on the World Health Organization's List of Essential Medicines, the most effective and safe medicines needed in a health system. The wholesale cost in the developing world is US$2.09 to US$2.57 a dose. It is not commercially available in the United States; however, it can be obtained via the Centers for Disease Control. It was originally made from the sweet wormwood plant. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
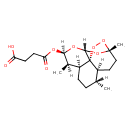
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Artesunic acid | Generator | | Dihydroartemisinine-12-alpha-succinate | MeSH | | Sodium artesunate | MeSH | | Dihydroartemisinine 12 alpha succinate | MeSH | | Succinyl dihydroartemisinin | MeSH | | Dihydroartemisinin, succinyl | MeSH | | Malartin | MeSH | | Malacef | MeSH | | Artesunate | MeSH | | 4-oxo-4-{[(1R,4S,5R,8S,9R,10R,12R,13R)-1,5,9-trimethyl-11,14,15,16-tetraoxatetracyclo[10.3.1.0⁴,¹³.0⁸,¹³]hexadecan-10-yl]oxy}butanoate | Generator |
|
|---|
| Chemical Formula | C19H28O8 |
|---|
| Average Molecular Mass | 384.425 g/mol |
|---|
| Monoisotopic Mass | 384.178 g/mol |
|---|
| CAS Registry Number | 88495-63-0 |
|---|
| IUPAC Name | 4-oxo-4-{[(1R,4S,5R,8S,9R,10R,12R,13R)-1,5,9-trimethyl-11,14,15,16-tetraoxatetracyclo[10.3.1.0⁴,¹³.0⁸,¹³]hexadecan-10-yl]oxy}butanoic acid |
|---|
| Traditional Name | 4-oxo-4-{[(1R,4S,5R,8S,9R,10R,12R,13R)-1,5,9-trimethyl-11,14,15,16-tetraoxatetracyclo[10.3.1.0⁴,¹³.0⁸,¹³]hexadecan-10-yl]oxy}butanoic acid |
|---|
| SMILES | [H][C@@]1(C)CC[C@@]2([H])[C@@]([H])(C)[C@@]([H])(OC(=O)CCC(O)=O)O[C@]3([H])O[C@@]4(C)CC[C@]1([H])[C@@]23OO4 |
|---|
| InChI Identifier | InChI=1S/C19H28O8/c1-10-4-5-13-11(2)16(23-15(22)7-6-14(20)21)24-17-19(13)12(10)8-9-18(3,25-17)26-27-19/h10-13,16-17H,4-9H2,1-3H3,(H,20,21)/t10-,11-,12+,13+,16+,17-,18-,19-/m1/s1 |
|---|
| InChI Key | FIHJKUPKCHIPAT-NKHDUEHSSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as artemisinins. These are sesquiterpenoids originally isolated from the herb Artemisia annua. Their structure is based on artemisinin, a tetracyclic compound that contains a 1,2-dioxepane fused to an octahydrobenzopyran moiety. The internal peroxide bridge is believed to be a key to the mode of action of artemisinins. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Sesquiterpenoids |
|---|
| Direct Parent | Artemisinins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Artemisinin skeleton
- Fatty acid ester
- Heterocyclic fatty acid
- Oxepane
- Dicarboxylic acid or derivatives
- Oxane
- Fatty acyl
- 1,2,4-trioxane
- Carboxylic acid ester
- Dialkyl peroxide
- Oxacycle
- Carboxylic acid
- Acetal
- Carboxylic acid derivative
- Organoheterocyclic compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Carbonyl group
- Organic oxygen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00kr-2459000000-709f5cba7340e7cb0802 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-7392000000-6aade85a0d3ce77db50e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4l-9050000000-c432eeabf339e3f7593b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000t-9034000000-4fd3316b534c1c9695e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00m0-4191000000-c1b630ac82bbe1ed3eec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-8590000000-1aec7778a60c3c83b96f | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Artesunate |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 65664 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|