| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 04:27:53 UTC |
|---|
| Update Date | 2016-11-09 01:15:37 UTC |
|---|
| Accession Number | CHEM017562 |
|---|
| Identification |
|---|
| Common Name | Xylazine |
|---|
| Class | Small Molecule |
|---|
| Description | A methyl benzene that is 1,3-dimethylbenzene which is substituted by a 5,6-dihydro-4H-1,3-thiazin-2-ylnitrilo group at position 2. It is an alpha2 adrenergic receptor agonist and frequently used in veterinary medicine as an emetic and sedative with analgesic and muscle relaxant properties. |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
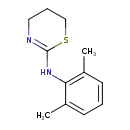
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-(2,6-Dimethylanilino)-5,6-dihydro-4H-1,3-thiazine | ChEBI | | 2-(2,6-Dimethylphenylamino)-4H-5,6-dihydro-1,3-thiazine | ChEBI | | 5,6-Dihydro-2-(2,6-xylidino)-4H-1,3-thiazine | ChEBI | | BAY 1470 | ChEBI | | Chanazine | ChEBI | | N-(5,6-Dihydro-4H-1,3-thiazinyl)-2,6-xylidine | ChEBI | | Rompun | ChEBI | | WH 7286 | ChEBI | | Xilazina | ChEBI | | Xilazine | ChEBI | | Xylazinum | ChEBI | | BAY va 1470 | MeSH | | BAY-va 1470 | MeSH | | BAYVa 1470 | MeSH | | Xylaxine | MeSH | | Xylazin | MeSH | | Xylazine | MeSH | | Xylazine hydrochloride | MeSH | | Xylazine monohydrochloride | MeSH | | Xylazine phosphate (1:1) | MeSH |
|
|---|
| Chemical Formula | C12H16N2S |
|---|
| Average Molecular Mass | 220.330 g/mol |
|---|
| Monoisotopic Mass | 220.103 g/mol |
|---|
| CAS Registry Number | 7361-61-7 |
|---|
| IUPAC Name | N-(2,6-dimethylphenyl)-5,6-dihydro-4H-1,3-thiazin-2-amine |
|---|
| Traditional Name | xylazine |
|---|
| SMILES | CC1=CC=CC(C)=C1NC1=NCCCS1 |
|---|
| InChI Identifier | InChI=1S/C12H16N2S/c1-9-5-3-6-10(2)11(9)14-12-13-7-4-8-15-12/h3,5-6H,4,7-8H2,1-2H3,(H,13,14) |
|---|
| InChI Key | BPICBUSOMSTKRF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as m-xylenes. These are aromatic compounds that contain a m-xylene moiety, which is a monocyclic benzene carrying exactly two methyl groups at the 1- and 3-positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Xylenes |
|---|
| Direct Parent | m-Xylenes |
|---|
| Alternative Parents | |
|---|
| Substituents | - M-xylene
- Aniline or substituted anilines
- Meta-thiazine
- Isothiourea
- Azacycle
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Organic nitrogen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-2920000000-828d7051a7615e6674a1 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-0229-3920000000-b822873abe0cc77b6500 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-0229-3920000000-b822873abe0cc77b6500 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-5190000000-4a2426f255239ce7000f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-2890000000-e9d742e119aa7bebbf65 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9100000000-874da3019fe4c2a17d31 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0159-1940000000-9ca9274ae36bfc2ed740 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03ka-2900000000-ab30d3e7c0d334cfbb33 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0c0c-9700000000-d509da0c01ee84ed9b7f | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB11477 |
|---|
| HMDB ID | HMDB0259938 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Xylazine |
|---|
| Chemspider ID | 5505 |
|---|
| ChEBI ID | 92386 |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|