| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 04:15:00 UTC |
|---|
| Update Date | 2016-11-09 01:15:34 UTC |
|---|
| Accession Number | CHEM017332 |
|---|
| Identification |
|---|
| Common Name | 4-Hydroxyphenyl retinamide |
|---|
| Class | Small Molecule |
|---|
| Description | A synthetic retinoid that is used orally as a chemopreventive against prostate cancer and in women at risk of developing contralateral breast cancer. It is also effective as an antineoplastic agent. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
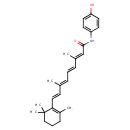
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 4-HPR | ChEBI | | 4-Hydroxy(phenyl)retinamide | ChEBI | | all-trans-4'-Hydroxyretinanilide | ChEBI | | Fenretinida | ChEBI | | Fenretinidum | ChEBI | | N-(4-Hydroxyphenyl)all-trans retinamide | ChEBI | | N-(4-Hydroxyphenyl)retinamide | ChEBI | | Fenretinide, 13-cis-isomer | MeSH | | 13-cis-Isomer fenretinide | MeSH | | 4 Hydroxyphenylretinamide | MeSH | | 4-Hydroxyphenylretinamide | MeSH | | Fenretinide, 13 cis isomer | MeSH | | MCN-R-1967 | MeSH | | Fenretinimide | MeSH | | MCN R 1967 | MeSH | | N-(4-Hydroxyphenyl)-trans-retinamide | MeSH |
|
|---|
| Chemical Formula | C26H33NO2 |
|---|
| Average Molecular Mass | 391.546 g/mol |
|---|
| Monoisotopic Mass | 391.251 g/mol |
|---|
| CAS Registry Number | 65646-68-6 |
|---|
| IUPAC Name | (2E,4E,6E,8E)-N-(4-hydroxyphenyl)-3,7-dimethyl-9-(2,6,6-trimethylcyclohex-1-en-1-yl)nona-2,4,6,8-tetraenamide |
|---|
| Traditional Name | fenretinide |
|---|
| SMILES | C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(=O)NC1=CC=C(O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C26H33NO2/c1-19(11-16-24-21(3)10-7-17-26(24,4)5)8-6-9-20(2)18-25(29)27-22-12-14-23(28)15-13-22/h6,8-9,11-16,18,28H,7,10,17H2,1-5H3,(H,27,29)/b9-6+,16-11+,19-8+,20-18+ |
|---|
| InChI Key | AKJHMTWEGVYYSE-FXILSDISSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as retinoids. These are oxygenated derivatives of 3,7-dimethyl-1-(2,6,6-trimethylcyclohex-1-enyl)nona-1,3,5,7-tetraene and derivatives thereof. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Retinoids |
|---|
| Direct Parent | Retinoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diterpenoid
- Retinoid skeleton
- Anilide
- N-arylamide
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Monocyclic benzene moiety
- Benzenoid
- Carboxamide group
- Secondary carboxylic acid amide
- Carboxylic acid derivative
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Organonitrogen compound
- Organic oxygen compound
- Carbonyl group
- Organic nitrogen compound
- Hydrocarbon derivative
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0889000000-39dd7388491d9985cc40 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03dr-0940000000-ac6e4f135e7a21a57933 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052r-2900000000-396248fb2a1713587c95 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0109000000-64792252df431097c271 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4l-0936000000-3ec8b35ee5dbc512fbf9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-3930000000-d8ce803307cd672ada48 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB05076 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Fenretinide |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 42588 |
|---|
| PubChem Compound ID | 5288209 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|