| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 04:14:50 UTC |
|---|
| Update Date | 2016-11-09 01:15:34 UTC |
|---|
| Accession Number | CHEM017329 |
|---|
| Identification |
|---|
| Common Name | Nocodazole |
|---|
| Class | Small Molecule |
|---|
| Description | A member of the class of benzimidazoles that is benzimidalole which is substituted at position 2 by a (methoxycarbonyl)amino group and at position 5 by a 2-thienoyl group. It is an antineoplastic agent that exerts its effect by depolymerising microtubules. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
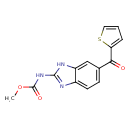
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (5-(2-Thienylcarbonyl)-1H-benzimidazol-2-yl)-carbamic acid methyl ester | ChEBI | | Methyl (5-(2-thienylcarbonyl))-1H-benzimidazole-2-ylcarbamate | ChEBI | | Methyl N-(5-thenoyl-2-benzimidazolyl)carbamate | ChEBI | | N-(5-(2-Thenoyl)-2-benzimidazolyl)carbamic acid methyl ester | ChEBI | | N-(5-(2-Thienoyl)-2-benzimidazolyl)carbamic acid methyl ester | ChEBI | | Nocodazol | ChEBI | | Nocodazolum | ChEBI | | Oncodazole | ChEBI | | R 17934 | ChEBI | | R-17934 | ChEBI | | R17,934 | ChEBI | | (5-(2-Thienylcarbonyl)-1H-benzimidazol-2-yl)-carbamate methyl ester | Generator | | Methyl (5-(2-thienylcarbonyl))-1H-benzimidazole-2-ylcarbamic acid | Generator | | Methyl N-(5-thenoyl-2-benzimidazolyl)carbamic acid | Generator | | N-(5-(2-Thenoyl)-2-benzimidazolyl)carbamate methyl ester | Generator | | N-(5-(2-Thienoyl)-2-benzimidazolyl)carbamate methyl ester | Generator |
|
|---|
| Chemical Formula | C14H11N3O3S |
|---|
| Average Molecular Mass | 301.320 g/mol |
|---|
| Monoisotopic Mass | 301.052 g/mol |
|---|
| CAS Registry Number | 31430-18-9 |
|---|
| IUPAC Name | methyl N-[6-(thiophene-2-carbonyl)-1H-1,3-benzodiazol-2-yl]carbamate |
|---|
| Traditional Name | nocodazole |
|---|
| SMILES | COC(=O)NC1=NC2=CC=C(C=C2N1)C(=O)C1=CC=CS1 |
|---|
| InChI Identifier | InChI=1S/C14H11N3O3S/c1-20-14(19)17-13-15-9-5-4-8(7-10(9)16-13)12(18)11-3-2-6-21-11/h2-7H,1H3,(H2,15,16,17,19) |
|---|
| InChI Key | KYRVNWMVYQXFEU-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzimidazoles. These are organic compounds containing a benzene ring fused to an imidazole ring (five member ring containing a nitrogen atom, 4 carbon atoms, and two double bonds). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzimidazoles |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzimidazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzimidazole
- Thiophene carboxylic acid or derivatives
- Aryl ketone
- Benzenoid
- Azole
- Imidazole
- Thiophene
- Heteroaromatic compound
- Ketone
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Azacycle
- Carboximidic acid derivative
- Organopnictogen compound
- Organic nitrogen compound
- Organic oxygen compound
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-03di-1790000000-825534d5d7b658640d20 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-00di-3790000000-449c2492c2ab67a70502 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0059000000-32c61dc92bf665194cfd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00kf-0190000000-0b03ee19763aafda8637 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-1490000000-b138a6f40b2cc9f1c17d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0ldi-4095000000-1acc188f83672b11d1c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-4092000000-a97f7670cc206c37f57c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9130000000-c16486ccbe15cf396319 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB08313 |
|---|
| HMDB ID | HMDB0255688 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Nocodazole |
|---|
| Chemspider ID | 3979 |
|---|
| ChEBI ID | 34892 |
|---|
| PubChem Compound ID | 4122 |
|---|
| Kegg Compound ID | C13719 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|