| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 04:00:40 UTC |
|---|
| Update Date | 2016-11-09 01:15:33 UTC |
|---|
| Accession Number | CHEM017199 |
|---|
| Identification |
|---|
| Common Name | 2,3,5,6-Tetrachloronitrobenzene |
|---|
| Class | Small Molecule |
|---|
| Description | A C-nitro compound that is nitrobenzene in which the four hydrogens located ortho- and para- to the nitro group have been replaced by chlorines. A fungicide used to control dry rot, it is no longer approved for use within the European Union. |
|---|
| Contaminant Sources | - FooDB Chemicals
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
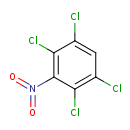
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2,3,5,6-TCNB | ChEBI | | 2,3,5,6-Tetrachlor-1-nitrobenzol | ChEBI | | 2,3,5,6-Tetrachloro-1-nitrobenzene | ChEBI | | 2,3,5,6-Tetrachloronitrobenzene | ChEBI | | Altritan | ChEBI | | Arena | ChEBI | | Bygran | ChEBI | | Easytec | ChEBI | | Folosan | ChEBI | | Fumite | ChEBI | | Fusarex | ChEBI | | Hickstor | ChEBI | | Hystore | ChEBI | | Myfusan | ChEBI | | Nebulin | ChEBI | | TCNB | ChEBI | | Tecnazen | ChEBI | | Tetrachloronitrobenzene | ChEBI | | Turbostore | ChEBI | | 1,2,4, 5-Tetrachloro-3-nitrobenzene | HMDB | | 1,2,4,5-Tetrachloro-3-nitro-benzene | HMDB | | 2,3,5, 6-Tetrachloro-1-nitrobenzene | HMDB | | 2,3,5,6-Tetrachlor-3-nitrobenzol | HMDB | | 3-Nitro-1,2,4,5-tetrachloro-benzene | HMDB | | Ashlade TCNB | HMDB | | Benzene, tetrachloronitro- (9ci) | HMDB | | Chipman 3,142 | HMDB | | Chipman 3142 | HMDB | | Folosan DB 905 | HMDB | | Folosan DB-905 | HMDB | | Fumite TCNB | HMDB | | Fumite TCNB smoke | HMDB | | Fumite techalin | HMDB | | Fusarex g | HMDB | | Fusarex t | HMDB | | Hystor | HMDB | | Hystor 10 | HMDB | | Hytec | HMDB | | Napotate | HMDB | | New hickstor 6 | HMDB | | New hystor | HMDB | | New hystore | HMDB | | Quad store | HMDB | | Quad-keep | HMDB | | Storite SS | HMDB | | Tecgran | HMDB | | Tecnazene, bsi, iso | HMDB | | Teknazen | HMDB | | Terraclor | HMDB | | Tripart arena 6 | HMDB | | Tripart arena granules | HMDB | | Tubodust | HMDB | | Tubostore | HMDB | | Tecnazine | HMDB |
|
|---|
| Chemical Formula | C6HCl4NO2 |
|---|
| Average Molecular Mass | 260.890 g/mol |
|---|
| Monoisotopic Mass | 258.876 g/mol |
|---|
| CAS Registry Number | 117-18-0 |
|---|
| IUPAC Name | 1,2,4,5-tetrachloro-3-nitrobenzene |
|---|
| Traditional Name | 1,2,4,5-tetrachloro-3-nitrobenzene |
|---|
| SMILES | ClC1=CC(Cl)=C(Cl)C(=C1Cl)N(=O)=O |
|---|
| InChI Identifier | InChI=1S/C6HCl4NO2/c7-2-1-3(8)5(10)6(4(2)9)11(12)13/h1H |
|---|
| InChI Key | XQTLDIFVVHJORV-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as nitrobenzenes. Nitrobenzenes are compounds containing a nitrobenzene moiety, which consists of a benzene ring with a carbon bearing a nitro group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Nitrobenzenes |
|---|
| Direct Parent | Nitrobenzenes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Nitrobenzene
- Nitroaromatic compound
- Chlorobenzene
- Halobenzene
- Aryl chloride
- Aryl halide
- C-nitro compound
- Organic nitro compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Allyl-type 1,3-dipolar organic compound
- Organic oxoazanium
- Organic nitrogen compound
- Organochloride
- Organohalogen compound
- Organonitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-08fr-0190000000-d8fa685fd7a55a4f0470 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0090000000-365c3aee9338cee48d34 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0090000000-2352f26a74cfa17544ac | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000t-1090000000-fd6ee0491271921e96b9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0090000000-99960be71288475e582d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0090000000-7218b34a10f0bbfc8e7f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4j-1390000000-fb2963dca35507d02795 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0090000000-f99a3dfe42a92720111d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0090000000-f99a3dfe42a92720111d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-0090000000-f99a3dfe42a92720111d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0090000000-4a410c9a08ca25950f33 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0090000000-4a410c9a08ca25950f33 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0090000000-4a410c9a08ca25950f33 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0031808 |
|---|
| FooDB ID | FDB008482 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 21106573 |
|---|
| ChEBI ID | 82044 |
|---|
| PubChem Compound ID | 8330 |
|---|
| Kegg Compound ID | C18897 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|