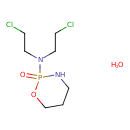
| (+-)-2-(Bis(2-chloroethyl)amino)tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide monohydrate | ChEBI |
| (Bis(chloro-2-ethyl)amino)-2-tetrahydro-3,4,5,6-oxazaphosphorine-1,3,2-oxide-2 monohydrate | ChEBI |
| 1-Bis(2-chloroethyl)amino-1-oxo-2-aza-5-oxaphosphoridine monohydrate | ChEBI |
| 2-(Bis(2-chloroethyl)amino)-1-oxa-3-aza-2-phosphocyclohexane 2-oxide monohydrate | ChEBI |
| 2-(Di(2-chloroethyl)amino)-1-oxa-3-aza-2-phosphacyclohexane-2-oxide monohydrate | ChEBI |
| Bis(2-chloroethyl)phosphoramide cyclic propanolamide ester monohydrate | ChEBI |
| Cyclophosphamide | ChEBI |
| Cyclophosphamide monohydrate | ChEBI |
| N,N-Bis(2-chloroethyl)tetrahydro-2H-1,3,2-oxaphosphorin-2-amine, 2-oxide monohydrate | ChEBI |
| N,N-Bis(beta-chloroethyl)-n',O-trimethylenephosphoric acid ester diamide monohydrate | ChEBI |
| Cytoxan | Kegg |
| Neosar | Kegg |
| (+-)-2-(Bis(2-chloroethyl)amino)tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide monohydric acid | Generator |
| (Bis(chloro-2-ethyl)amino)-2-tetrahydro-3,4,5,6-oxazaphosphorine-1,3,2-oxide-2 monohydric acid | Generator |
| 1-Bis(2-chloroethyl)amino-1-oxo-2-aza-5-oxaphosphoridine monohydric acid | Generator |
| 2-(Bis(2-chloroethyl)amino)-1-oxa-3-aza-2-phosphocyclohexane 2-oxide monohydric acid | Generator |
| 2-(Di(2-chloroethyl)amino)-1-oxa-3-aza-2-phosphacyclohexane-2-oxide monohydric acid | Generator |
| Bis(2-chloroethyl)phosphoramide cyclic propanolamide ester monohydric acid | Generator |
| Cyclophosphamide monohydric acid | Generator |
| N,N-Bis(2-chloroethyl)tetrahydro-2H-1,3,2-oxaphosphorin-2-amine, 2-oxide monohydric acid | Generator |
| N,N-Bis(b-chloroethyl)-n',O-trimethylenephosphate ester diamide monohydrate | Generator |
| N,N-Bis(b-chloroethyl)-n',O-trimethylenephosphoric acid ester diamide monohydric acid | Generator |
| N,N-Bis(beta-chloroethyl)-n',O-trimethylenephosphate ester diamide monohydrate | Generator |
| N,N-Bis(beta-chloroethyl)-n',O-trimethylenephosphoric acid ester diamide monohydric acid | Generator |
| N,N-Bis(β-chloroethyl)-n',O-trimethylenephosphate ester diamide monohydrate | Generator |
| N,N-Bis(β-chloroethyl)-n',O-trimethylenephosphoric acid ester diamide monohydric acid | Generator |
| Cyclophosphamide hydric acid | Generator |
| N,N-Bis(2-chloroethyl)-2-oxo-1,3,2$l^{5}-oxazaphosphinan-2-amine;hydric acid | Generator |
| Cyclophosphane | MeSH |
| Monohydrate, cyclophosphamide | MeSH |
| Cyclophosphamide anhydrous | MeSH |
| Cyclophosphamide, (S)-isomer | MeSH |
| Cyclophosphamide, (R)-isomer | MeSH |
| Cytophosphan | MeSH |
| Procytox | MeSH |
| Sendoxan | MeSH |
| Cytophosphane | MeSH |
| Anhydrous, cyclophosphamide | MeSH |
| Endoxan | MeSH |
| Cyclophosphamide hydrate | KEGG |
| Cyclophosphamide tablets | KEGG |
| (+,-)-2-(Bis(2-chloroethyl)amino)tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide monohydrate | MeSH |