| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:48:27 UTC |
|---|
| Update Date | 2016-11-09 01:15:30 UTC |
|---|
| Accession Number | CHEM016947 |
|---|
| Identification |
|---|
| Common Name | Saquinavir mesylate |
|---|
| Class | Small Molecule |
|---|
| Description | Saquinavir, sold under the brand names Invirase and Fortovase, is an antiretroviral drug used together with other medications to treat or prevent HIV/AIDS. Typically it is used with ritonavir or lopinavir/ritonavir to increase its effect. It is taken by mouth.Common side effects include nausea, vomiting, diarrhea, and feeling tired. More serious side effects include problems with QT prolongation, heart block, high blood lipids, and liver problems. It appears to be safe in pregnancy. It is in the protease inhibitor class and works by blocking the HIV protease.Saquinavir was patented in 1988 and first sold in 1995. As of 2015 it is not available as a generic medication in the United States and is expensive. The wholesale cost in the developing world is about 4.50 USD per day. |
|---|
| Contaminant Sources | - ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
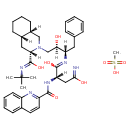
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Saquinavir mesilate | Kegg | | Invirase | Kegg | | Saquinavir mesilic acid | Generator | | Saquinavir mesylic acid | Generator | | Saquinavir monomethanesulfonate | MeSH | | Monomethanesulfonate, saquinavir | MeSH | | Saquinavir | MeSH | | Saquinivir | MeSH |
|
|---|
| Chemical Formula | C39H54N6O8S |
|---|
| Average Molecular Mass | 766.960 g/mol |
|---|
| Monoisotopic Mass | 766.372 g/mol |
|---|
| CAS Registry Number | 149845-06-7 |
|---|
| IUPAC Name | (2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butyl-C-hydroxycarbonimidoyl)-decahydroisoquinolin-2-yl]-3-hydroxy-1-phenylbutan-2-yl]-2-[(quinolin-2-yl)formamido]butanediimidic acid; methanesulfonic acid |
|---|
| Traditional Name | invirase; methanesulfonic acid |
|---|
| SMILES | CS(O)(=O)=O.[H][C@@](O)(CN1C[C@@]2([H])CCCC[C@@]2([H])C[C@@]1([H])C(O)=NC(C)(C)C)[C@]([H])(CC1=CC=CC=C1)N=C(O)[C@]([H])(CC(O)=N)NC(=O)C1=NC2=CC=CC=C2C=C1 |
|---|
| InChI Identifier | InChI=1S/C38H50N6O5.CH4O3S/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29;1-5(2,3)4/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49);1H3,(H,2,3,4)/t26-,27+,30-,31-,32-,33+;/m0./s1 |
|---|
| InChI Key | IRHXGOXEBNJUSN-YOXDLBRISA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as chlorohydrins. These are alcohols substituted by a chlorine atom at a saturated carbon atom otherwise bearing only hydrogen or hydrocarbyl groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organohalogen compounds |
|---|
| Class | Halohydrins |
|---|
| Sub Class | Chlorohydrins |
|---|
| Direct Parent | Chlorohydrins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Chlorohydrin
- Organic oxygen compound
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Organochloride
- Alkyl halide
- Alkyl chloride
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000000900-0cf34e5ca3aa1cabaa8a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0000000900-0cf34e5ca3aa1cabaa8a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-0000000900-0cf34e5ca3aa1cabaa8a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0000000900-c5b70513d21499cd5a1a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0000000900-c5b70513d21499cd5a1a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014i-0000000900-c5b70513d21499cd5a1a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Saquinavir |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 60934 |
|---|
| Kegg Compound ID | C12560 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|