| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:48:04 UTC |
|---|
| Update Date | 2016-11-09 01:15:29 UTC |
|---|
| Accession Number | CHEM016934 |
|---|
| Identification |
|---|
| Common Name | Vinpocetine |
|---|
| Class | Small Molecule |
|---|
| Description | Vinpocetine has been investigated for the treatment of Epilepsy. |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Kavinton | MeSH | | Ethyl apovincaminate, (16alpha)-isomer | MeSH | | Cavinton | MeSH | | Ethyl apovincaminate | MeSH | | Ethyl apovincaminate, 3H-labeled | MeSH | | TCV-3b | MeSH | | Ethyl apovincaminate tartrate (1:1) | MeSH | | Ethyl apovincaminate, (+-)-isomer | MeSH | | Ethyl apovincaminate, 2-oxopentanedioate (1:1) | MeSH | | Ethyl apovincaminate, (3alpha,16alpha)-isomer | MeSH | | AY-27255vinpocetine | ChEMBL |
|
|---|
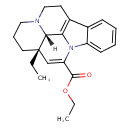
| Chemical Formula | C22H26N2O2 |
|---|
| Average Molecular Mass | 350.462 g/mol |
|---|
| Monoisotopic Mass | 350.199 g/mol |
|---|
| CAS Registry Number | 42971-09-5 |
|---|
| IUPAC Name | ethyl (15S,19S)-15-ethyl-1,11-diazapentacyclo[9.6.2.0²,⁷.0⁸,¹⁸.0¹⁵,¹⁹]nonadeca-2,4,6,8(18),16-pentaene-17-carboxylate |
|---|
| Traditional Name | vinpocetine |
|---|
| SMILES | [H][C@]12N3CCC[C@@]1(CC)C=C(N1C4=CC=CC=C4C(CC3)=C21)C(=O)OCC |
|---|
| InChI Identifier | InChI=1S/C22H26N2O2/c1-3-22-11-7-12-23-13-10-16-15-8-5-6-9-17(15)24(19(16)20(22)23)18(14-22)21(25)26-4-2/h5-6,8-9,14,20H,3-4,7,10-13H2,1-2H3/t20-,22+/m1/s1 |
|---|
| InChI Key | DDNCQMVWWZOMLN-IRLDBZIGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as eburnan-type alkaloids. These are alkaloids with a structure based on the eburnan skeleton, that arises from rearrangement of the aspidospermidine ring system, involving migration of C-21 from C-7 to C-2, fission of the 2,16-bond, and attachment of C-16 to N-1. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Eburnan-type alkaloids |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Eburnan-type alkaloids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Eburna alkaloid
- Indolo[3,2-1de][1,5]naphthyridine
- Beta-carboline
- Pyridoindole
- Diazanaphthalene
- Alpha-amino acid or derivatives
- Naphthyridine
- 3-alkylindole
- Indole
- Indole or derivatives
- Aralkylamine
- Piperidine
- Benzenoid
- Heteroaromatic compound
- Pyrrole
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- Amino acid or derivatives
- Tertiary aliphatic amine
- Tertiary amine
- Carboxylic acid ester
- Organoheterocyclic compound
- Azacycle
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Amine
- Organic oxide
- Organopnictogen compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Carbonyl group
- Organic oxygen compound
- Organonitrogen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-0fsi-1292000000-d2a82dd6f1fdac32445e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0009000000-8425286cda67c150b9a3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0pdi-1049000000-5ef2be9ce63e54b48e54 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-06vi-0093000000-15e087d4e3f1ce581539 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0f6t-1009000000-255dcda558a1da91c36a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f92-3049000000-ccfc94de1322e79544fe | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-002b-6094000000-661d6360f8ead23bfef9 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB12131 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Vinpocetine |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 443955 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|