| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-22 03:47:23 UTC |
|---|
| Update Date | 2026-03-26 23:07:47 UTC |
|---|
| Accession Number | CHEM016912 |
|---|
| Identification |
|---|
| Common Name | Tiopronin |
|---|
| Class | Small Molecule |
|---|
| Description | Tiopronin is a prescription thiol drug used primarily in the treatment of severe homozygous cystinuria. Patients with cystinuria excrete high levels of cystine in their urine and are at risk for kidney stone formation. Tiopronin is used as a second-line therapy to control the rate of cystine precipitation and excretion, and prevent kidney stone formation. It is used after a failure of the non-pharmacological first line treatment consisting of increased fluid intake, restriction of sodium and protein, and urinary alkalinization. As cystinuria is a relatively rare disease, tiopronin is classified as an orphan drug and is not patented in the United States. It is similar to d-penicillamine in use and efficacy, but offers the advantage of far less adverse effects. Tiopronin is dosed on an individual basis using close monitoring of urinary cystine concentrations and urinary output.
Tiopronin may also be used to bind metal nanoparticles in Wilson's disease, which is an overload of copper in the body. It has been investigated for use in the treatment of arthritis and as a neuroprotective agent in aneurysmal subarachnoid hemorrhage. |
|---|
| Contaminant Sources | - STOFF IDENT Compounds
- ToxCast & Tox21 Chemicals
|
|---|
| Contaminant Type | Not Available |
|---|
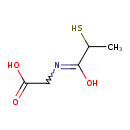
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Thiola | Kegg | | 2-(2-Sulfanylpropanoylamino)acetate | Generator | | 2-(2-Sulphanylpropanoylamino)acetate | Generator | | 2-(2-Sulphanylpropanoylamino)acetic acid | Generator | | 2 Mercaptopropionylglycine | MeSH | | 2 Thiol propionamido acetic acid | MeSH | | 2 Thiolpropionamidoacetic acid | MeSH | | 2-Mercaptopropionylglycine | MeSH | | 2-Thiol-propionamido-acetic acid | MeSH | | 2-Thiolpropionamidoacetic acid | MeSH | | Acadione | MeSH | | Acid, 2-thiol-propionamido-acetic | MeSH | | Acid, 2-thiolpropionamidoacetic | MeSH | | Aventis brand OF tiopronin | MeSH | | Captimer | MeSH | | MIT gesundheit brand OF tiopronin | MeSH | | Meprin | MeSH | | Mercaptopropionylglycine | MeSH | | Thiopronine | MeSH | | Tiopronin | MeSH | | Tiopronin aventis brand | MeSH | | Tiopronine | MeSH | | alpha Mercaptopropionylglycine | MeSH | | alpha-Mercaptopropionylglycine | MeSH |
|
|---|
| Chemical Formula | C5H9NO3S |
|---|
| Average Molecular Mass | 163.190 g/mol |
|---|
| Monoisotopic Mass | 163.030 g/mol |
|---|
| CAS Registry Number | 1953-02-2 |
|---|
| IUPAC Name | 2-[(1-hydroxy-2-sulfanylpropylidene)amino]acetic acid |
|---|
| Traditional Name | capen |
|---|
| SMILES | CC(S)C(O)=NCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C5H9NO3S/c1-3(10)5(9)6-2-4(7)8/h3,10H,2H2,1H3,(H,6,9)(H,7,8) |
|---|
| InChI Key | YTGJWQPHMWSCST-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as n-acyl-alpha amino acids. N-acyl-alpha amino acids are compounds containing an alpha amino acid which bears an acyl group at its terminal nitrogen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | N-acyl-alpha amino acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - N-acyl-alpha-amino acid
- Carboxamide group
- Secondary carboxylic acid amide
- Alkylthiol
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic nitrogen compound
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0c2l-9200000000-c5efdfa9a89aebdce73f | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_2_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_2_3) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03k9-6900000000-2be39fc38fff76940bf7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00fr-9000000000-6b589fda01eac68ee080 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00b9-9000000000-e3fcfcc65e679e9ca168 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-3900000000-092a4bb4dee549564be0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03kc-5900000000-20328de1666ad6c7e471 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05fr-9100000000-693c909a05b979f3e120 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB06823 |
|---|
| HMDB ID | HMDB0259104 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Tiopronin |
|---|
| Chemspider ID | 5283 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | C12876 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|