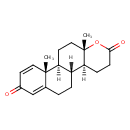
Identification Common Name Testolactone Class Small Molecule Description An antineoplastic agent that is a derivative of progesterone and used to treat advanced breast cancer. Contaminant Sources HMDB Contaminants - Urine
STOFF IDENT Compounds
ToxCast & Tox21 Chemicals Contaminant Type Not Available Chemical Structure Synonyms Value Source (4AS,4BR,10ar,10BS,12as)-10a,12a-dimethyl-3,4,4a,5,6,10a,10b,11,12,12a-decahydro-2H-naphtho[2,1-F]chromene-2,8(4BH)-dione ChEBI 1,2-Didehydrotestololactone ChEBI 1-Dehydrotestololactone ChEBI 13-Hydroxy-3-oxo-13,17-secoandrosta-1,4-dien-17-oic acid delta-lactone ChEBI D-homo-17a-Oxaandrosta-1,4-diene-3,17-dione ChEBI delta(1)-Testololactone ChEBI Teslac ChEBI 13-Hydroxy-3-oxo-13,17-secoandrosta-1,4-dien-17-oate delta-lactone Generator 13-Hydroxy-3-oxo-13,17-secoandrosta-1,4-dien-17-oate Δ-lactone Generator 13-Hydroxy-3-oxo-13,17-secoandrosta-1,4-dien-17-oic acid Δ-lactone Generator Δ(1)-testololactone Generator 1-Dehydrotestolactone MeSH, HMDB 1 Dehydrotestolactone MeSH, HMDB delta(1)-Testolactone MeSH, HMDB 1,2-Dehydrotestololactone HMDB Testolactone HMDB delta1-Dehydrotestololactone HMDB delta1-Testolactone HMDB delta1-Testololactone HMDB Δ1-Dehydrotestololactone HMDB Δ1-Testolactone HMDB Δ1-Testololactone HMDB
Chemical Formula C19 H24 O3 Average Molecular Mass 300.392 g/mol Monoisotopic Mass 300.173 g/mol CAS Registry Number 968-93-4 IUPAC Name (1R,2S,7S,10S,11R)-7,11-dimethyl-6-oxatetracyclo[8.8.0.0^{2,7}.0^{11,16}]octadeca-12,15-diene-5,14-dione Traditional Name (1R,2S,7S,10S,11R)-7,11-dimethyl-6-oxatetracyclo[8.8.0.0^{2,7}.0^{11,16}]octadeca-12,15-diene-5,14-dione SMILES [H][C@@]12CCC3=CC(=O)C=C[C@]3(C)[C@@]1([H])CC[C@]1(C)OC(=O)CC[C@@]21[H] InChI Identifier InChI=1S/C19H24O3/c1-18-9-7-13(20)11-12(18)3-4-14-15(18)8-10-19(2)16(14)5-6-17(21)22-19/h7,9,11,14-16H,3-6,8,10H2,1-2H3/t14-,15+,16+,18+,19+/m1/s1 InChI Key BPEWUONYVDABNZ-DZBHQSCQSA-N